Virtual Library
Start Your Search
Nicholas R. Faris
Author of
-
+
MA25 - Oligometastasis: Defining, Treating, and Evaluating (ID 929)
- Event: WCLC 2018
- Type: Mini Oral Abstract Session
- Track: Oligometastatic NSCLC
- Presentations: 1
- Moderators:
- Coordinates: 9/26/2018, 13:30 - 15:00, Room 203 BD
-
+
MA25.05 - Characteristics & Survival of Resected Stage IV Non-Small Cell Lung Cancer (NSCLC) in the Mid-South Quality of Surgical Resection Cohort (ID 13381)
14:00 - 14:05 | Presenting Author(s): Nicholas R. Faris
- Abstract
- Presentation
Background
Surgical resection is potentially curative in subsets of oligometastatic NSCLC. We evaluated the characteristics and survival of resected stage IV NSCLC in a population-based cohort.
a9ded1e5ce5d75814730bb4caaf49419 Method
Patients were included who had curative-intent resections from 11 hospitals in 4 contiguous Dartmouth Hospital Referral Regions in the mid-Southern USA from 2009-2018. Statistical analyses were performed using univariate and multiple Cox regression models.
4c3880bb027f159e801041b1021e88e8 Result
Of 3092 resections, 96 (3.1%) were stage IV: 38 M1a, 54 M1b, and 4 M1c. Of the M1a patients, 1 had a pleural effusion, 37 had a contralateral lung nodule. The most common sites of extrathoracic metastasis were bone (13 (13.5%)), and brain (25 (26%)). Other extrathoracic sites were distant lymph nodes, liver, adrenals, thyroid, pancreas, colon, soft tissue, and esophagus.
Stage IV patients had a younger median age (63 vs 67 (p<0.0001)), less Medicare coverage but more Medicaid or Commercial insurance (p=0.0248), fewer comorbid conditions (p=0.0096), higher cT (p<0.0001), and higher-grade tumors (p=0.0002).
58% (22) of M1a patients did not receive treatment to the site of metastatic disease, compared to 72% (39) and 75% (3) of M1b and M1c, respectively (p=0.0086).
For patients with bone metastases, median/5 year survival was 1.28 years/0%, compared to 5.16 years/51% for all other metastatic sites and 6.39 years/56% for non-stage IV NSCLC (p=0.0058) (Figure 1). In fully adjusted models, survival for Stage IV patients without bone metastasis did not differ significantly from Stage I-III patients (HR: 1.3, p=0.15). However, Stage IV patients with bone metastasis had significantly worse survival (HR:3.2, p=0.0006).
8eea62084ca7e541d918e823422bd82e Conclusion
Bone metastasis connotes a very poor prognosis in patients with oligometastatic NSCLC, but survival of patients with other sites of metastasis was remarkably good in this highly selected group of patients from a population-based multi-institutional cohort.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
P1.11 - Screening and Early Detection (Not CME Accredited Session) (ID 943)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.11-11 - Comparing Lung Cancer Diagnosed by Low Dose CT (LDCT), Incidental Lung Nodule Program (ILNP), and Non-Program-Based Detection (ID 14379)
16:45 - 18:00 | Author(s): Nicholas R. Faris
- Abstract
Background
The aggregate 5-year survival of lung cancer patients is <20%, partly because most patients present with advanced disease. LDCT screening and algorithmic management of patients with incidentally-detected nodules are two methods for early detection, but rigorous evaluation is needed for effective implementation. We compared patients with lung cancer diagnosed via LDCT vs. ILNP vs. neither in a lung cancer-endemic US region.
a9ded1e5ce5d75814730bb4caaf49419 Method
We compared demographic, clinical, and treatment characteristics of patients diagnosed via LDCT and ILNP with those treated in a multidisciplinary program (MDP) who were not diagnosed through either early detection program. LDCT screening was implemented in 2015 using Medicare eligibility criteria. In the ILNP, navigators and a multidisciplinary team prospectively tracked patients with suspicious findings flagged by radiologists, using Natural Language Processing software. All patients were diagnosed within the same healthcare system from 2015-2018. Statistical comparisons used chi-square, Fishers Exact, and ANOVA.
4c3880bb027f159e801041b1021e88e8 Result
Lung cancer diagnoses included 111 from 5,954 ILNP scans, 11 from 400 LDCT scans (1.9% v 2.8%, p=0.21), and 273 from MDC. An additional 40 (10%) LDCT scans were Lung RADS 3 or 4. Average ages were 70/68/68 years for ILNP/LDCT/MDC and patients were 43%/64%/48% male. African Americans were underrepresented in both early detection groups (23%/9%/36%; p=0.0111); Medicare patients were over-represented (83%/91%/42%, p<0.001). Active smoking was highest in LDCT (73%, 79 pack-year average), but similar between ILNP and MDC (39%, 50 pack-year average vs 36%, 63 pack-year average).
Early detection cases were more frequently adenocarcinoma (61%/ 55%/48%; p=0.0595) with smaller lesions (2.2cm/1.0cm/4.2cm; p<0.001). Stage I/II cancers were more likely with early detection (71%/89%/42%; p<0.001), leading to substantially higher rates of surgical resection (75%/73%/31%; p<0.001). Median time from lesion detection to treatment initiation was similar between groups (61/74/58 days, p=0.48).
62% of patients with lung cancers diagnosed by ILNP and 57% by MDC were not eligible for LDCT screening. The most common disqualifying criteria were a 30 pack-year smoking history (unmet: 41% ILNP/ 41% MDC) and active smoking within 15 years (unmet: 41% ILNP/ 27% MDC).
8eea62084ca7e541d918e823422bd82e Conclusion
Lung cancers diagnosed by ILNP and LDCT had better prognosis than the MDC population, with smaller tumors, earlier stage, and more treatment with surgical resection. African Americans were underrepresented in these groups. Less than half of ILNP and MDC cases had enough smoking exposure to qualify for LDCT screening. ILNP is a critical component of early detection programs, reaching an element of the population that did not qualify for LDCT screening.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P1.15 - Treatment in the Real World - Support, Survivorship, Systems Research (Not CME Accredited Session) (ID 947)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.15-21 - Creating an Optimal Care Coordination Model to Improve Multidisciplinary Care for Lung Cancer Patients on Medicaid (ID 12318)
16:45 - 18:00 | Author(s): Nicholas R. Faris
- Abstract
Background
The Association of Community Cancer Centers (ACCC) created an Optimal Care Coordination Model (OCCM), which provides a comprehensive self-assessment tool designed to orient cancer programs to achieving patient-centered, multidisciplinary care. The OCCM is designed to help cancer programs, regardless of resources, location, or population, improve care for lung cancer patients, especially those on Medicaid.
a9ded1e5ce5d75814730bb4caaf49419 Method
Using findings from an environmental scan (April 2016) and visits to 5 US cancer programs to explore current care models (July-October 2016), a Technical Expert Panel developed the OCCM, which has 13 defined Assessment Areas and utilizes an evaluation matrix (Table 1).
To validate the OCCM, a competitive application process among ACCC’s membership used a comprehensive institutional quantitative and qualitative questionnaire. Applicants completed a self-assessment using the OCCM and then developed quality improvement projects designed to move their OCCM-scored care delivery performance from baseline to a higher level over a 12-month implementation period. Seven US community cancer centers were selected as Testing Sites. Quantifiable outcome measures were identified for each site, standardized across sites, and collected by a centralized data coordinating center.
4c3880bb027f159e801041b1021e88e8 ResultTable 1
OCCM Assessment Areas
1. Patient Access to Care
8. Survivorship Care
2. Prospective Multidisciplinary Case Planning
9. Supportive Care
3. Financial, Transportation, and Housing
10. Tobacco Cessation
4. Management of Comorbid Conditions
11. Clinical Trials
5. Care Coordination
12. Physician Engagement
6. Treatment Team Integration
13. Quality Measurement and Improvement
7. Electronic Health Records and Patient Access to Information
Level 1:
Optimal care coordination for lung cancer care has a low priority as evidenced by fragmented care.
Level 2:
Early progress in coordinating care is underway.
Level 3:
Reflects average or typical care coordination.
Level 4:
Exceeds the average and reflects a cancer program’s ongoing commitment to the pursuit of optimal care coordination.
Level 5:
Defined by optimal care coordination with a patient-centered focus. Depending on the assessment area, achieving Level 5 performance will require significant time, effort, and resources.
Patient Focus:
Optimal care coordination must be patient-centered, which requires understanding of what is important to patients and their caregivers, including their knowledge, goals, needs, desires, social connections, and resources for care. This requires the cancer program to educate and engage patients and caregivers to facilitate shared decision-making and patients’ participation in their care.
Quality Measures and Metrics:
Each assessment area requires at least one measurable parameter. Optimal care coordination requires analysis and development of an action plan for continuous improvement. These parameters should include both evidence-based and institution-specific benchmarks that address patient outcomes, patient experience, and cost effectiveness. These measures and metrics should be continuously measured and fed back to key institutional stakeholders for ongoing quality improvement.
Table 2 shows Assessment Areas being validated and patient demographics.
8eea62084ca7e541d918e823422bd82e ConclusionTable 2
Site 1
Site 2
Site 3
Site 4
Site 5
Site 6
Site 7
TOTAL
Project # 1 Selected Assessment Area(s)
#2
#7
#10
#2
#1
#5 & #9
#1
Project # 2 Selected Assessment Area
#8
#11
n/a
#10
n/a
n/a
#2
8 of 13 Assessment Areas being validated
N=50
N=29
N=77
N=53
N=76
N=101
N=35
N=421
Age
Median
(IQR)
70
(57-76)
74
(63-76)
71
(57-76)
65
(60-71)
68
(61-75)
61
(55-65)
66
(60-73)
68
(61-74)
n (%)
n (%)
n (%)
n (%)
n (%)
n (%)
n (%)
n (%)
Sex
Male
29 (58)
14 (48)
40 (52)
27 (51)
49 (64)
48 (48)
18 (51)
225 (53)
Female
21 (42)
15 (52)
37 (48)
26 (49)
27 (36)
52 (51)
17 (49)
195 (46)
Race
Caucasian
46 (92)
23 (79)
76 (99)
51 (96)
73 (96)
47 (47)
27 (77)
343 (81)
African American
4 (8)
1 (3)
2 (4)
5 (5)
6 (17)
18 (4)
Asian
4 (14)
8 (8)
12 (3)
Other/
Unknown
1(1)
1 (1)
8(8)
2 (6)
12 (3)
Not reported
1 (3)
2 (3)
29 (29)
32 (8)
Insurance
Commercial
7 (14)
7 (24)
13 (17)
8 (15)
29 (38)
8 (8)
9 (26)
81 (19)
Medicare
38 (76)
20 (69)
55 (71)
37 (70)
40 (53)
28 (28)
21 (60)
239 (57)
Medicaid
4 (8)
2 (7)
6 (8)
8 (15)
6 (8)
63 (62)
4 (11)
93 (22)
None/Self-Pay
2 (3)
1 (1)
3 (1)
Smoking Status
Active Smoker
14 (28)
10 (34)
27 (35)
17 (32)
38 (50)
34 (34)
11 (31)
151 (36)
Former Smoker
20 (40)
15 (52)
44 (57)
34 (64)
28 (37)
51 (50)
23 (66)
215 (51)
Never Smoker
7 (14)
3 (10)
2 (3)
2 (4)
7 (9)
9 (9)
30 (7)
Not reported
2 (3)
1 (1)
3 (1)
Stage at Diagnosis
Stage 0
2 (3)
2 (<1)
Stage I
9 (18)
4 (14)
21 (27)
1 (2)
9 (12)
9 (9)
2 (6)
55 (13)
Stage II
1 (2)
2 (7)
10 (13)
5 (9)
3 (4)
7 (7)
28 (7)
Stage III
6 (12)
5 (17)
11 (14)
3 (6)
4 (5)
13 (13)
2 (6)
44 (10)
Stage IV
6 (12)
3 (10)
8 (10)
14 (26)
13 (17)
18 (18)
4 (11)
66 (16)
Not reported/ Missing
28 (56)
15 (52)
25 (32)
30 (57)
47 (62)
54 (53)
27 (77)
226 (54)
*Percentages may not add to 100 due to missing data
Project implementation and patient accrual are ongoing at all Testing Sites through September 2018.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P1.16 - Treatment of Early Stage/Localized Disease (Not CME Accredited Session) (ID 948)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.16-46 - A Population-Based Validation Study of the Proposed ‘R-Factor’ Classification in a Lung Cancer-Endemic Region of the US. (ID 13985)
16:45 - 18:00 | Author(s): Nicholas R. Faris
- Abstract
Background
The IASLC has proposed a definition of completeness of surgical resection beyond margin status. We sought to validate the proposed classification in a US cohort, and evaluated the impact of a lymph node (LN) specimen collection kit on resection status.
a9ded1e5ce5d75814730bb4caaf49419 Method
The population-based Mid-South Quality of Surgical Resection cohort includes >95% of lung cancer resections in 4 contiguous US Dartmouth Hospital Referral Regions from 2009-2018. Resections were classified as Complete (R0), Uncertain (R[un]), or Incomplete (R1-R2) based on the proposed classifications. We evaluated overall survival (OS) using the Kaplan-Meier method and proportional hazards models. Adjusted models included age, sex, histology, extent of resection, pTNM categories, and co-morbidities. A subset of resections used a LN specimen collection kit.
4c3880bb027f159e801041b1021e88e8 Result
Of 3,099 resections, 18% were R0, 76% R(un), and 6% R1-R2. 5-year OS was 69%/54%/35% for R0/R(un)/R1-R2 (p<0.0001, Figure 1A). Compared to R0, the increased hazard of death for R(un)/R1-R2 was 1.6/3.0 overall, 1.5/2.3 in node negative patients, 1.7/3.1 in node positive patients, and 1.5/1.9 in fully adjusted models (all p<0.0001).
Of 2,351 R(un) resections, the highest mediastinal LN positive increased the hazard of death 1.6 times (vs. negative, p=0.0008). However, 626 (27%) had no mediastinal LN examined (MLE). R(un) resections with 0 MLE had 1.2 times the hazard of death compared to R(un) with >1 MLE (p=0.0212, Figure 1B).
Use of the LN kit intervention resulted in R0 in 40% of cases, compared to 6% without the kit (p<0.0001). Kit cases had improved OS across the entire cohort (p=0.0002), but when restricted to R0 patients, OS did not differ based on kit use (p=0.96).
8eea62084ca7e541d918e823422bd82e Conclusion
The proposed ‘R-factor’ classifications are prognostic. R(un) rates were high, but significantly lower in cases where a LN collection kit was used. Further delineation of R(un) cases based on MLE should be considered.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P1.17 - Treatment of Locoregional Disease - NSCLC (Not CME Accredited Session) (ID 949)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.17-11 - Pattern and Survival Impact of Neoadjuvant Treatment of Non-Small Cell Lung Cancer (NSCLC) in a Prospective Lung Resection Cohort (ID 14169)
16:45 - 18:00 | Author(s): Nicholas R. Faris
- Abstract
Background
Neoadjuvant therapy may benefit locally-advanced NSCLC patients. We evaluated patterns of neoadjuvant therapy and the impact on stage-shift and survival.
a9ded1e5ce5d75814730bb4caaf49419 Method
All curative-intent NSCLC resections were collected from 12 hospitals in 4 contiguous Dartmouth Hospital Referral Regions in mid-Southern USA from 2009-2018. Comparisons made using Chi-square tests and non-parametric t-tests, survival impact assessed using Cox-proportional hazard models.
4c3880bb027f159e801041b1021e88e8 Result
182 of 3,297 resections (5.5%) had neoadjuvant therapy: 118 (64.8%) chemoradiation, 47 (25.8%) chemotherapy, and 17 (9.3%) radiation. Neoadjuvantly treated patients were younger, more likely to be commercially insured, have immediate preoperative brain MRI, and invasive mediastinal staging than those with primary resection (p<0.001 for all, Table 1). They also had more advanced stage, but 27% were clinical stage IA/IB.
Despite evidence of more difficult surgery, perioperative complications, hospital length of stay and postoperative mortality rates were similar to the primary resection cohort. Despite delay to surgery, they had significantly greater down-staging (p<0.001). However, down-staging had no impact on survival, regardless of type of neoadjuvant therapy (Table 2).
Table 1. Demographic and clinical characteristics between patients who received neoadjuvant therapy and those who did not. Demographic Variables
Neoadjuvant Therapy
Primary Resection Only
N
N=182
N=3115
Race (p: 0.0324)
Caucasian
125 (69)
2425 (78)
Black or AA
56 (31)
651 (21)
Asian
0 (0)
16 (1)
AI/AN
0 (0)
2 (0)
Other/NR
1 (1)
19 (1)
Age (p: <0.001)
63 (56, 69)
68 (61, 74)
Sex (p: 0.4323)
Male
104 (57)
1686 (54)
Female
78 (43)
1427 (46)
Insurance (p: <0.001)
Medicare
45 (25)
1417 (45)
Medicaid
42 (23)
438 (14)
Commercial
91 (50)
1148 (37)
Self/ None
4 (2)
112 (4)
Non-Invasive Staging
CT Scan ϯ
135 (74)
2879 (92)
PET/CT ϯ
106 (58[RO2] )
2553 (82)
Brain Scan (p: <0.001)
98 (54)
877 (28)
Invasive staging tests (p: <0.001)
No
144 (79)
2778 (89)
Yes
38 (21)
337 (11)
Histology (p: <0.001)
Adenocarcinoma
65 (40)
1547 (54)
Squamous
57 (35)
985 (34)
Other including but limited to Adenosqamous, large cell, carcinomas, and other
40 (22)
339 (11)
Grade (p: <0.001)
Well/Moderately
50 (30)
1684 (54)
Poorly/Undifferentiated
60 (33)
1040 (34)
Not Reported
66 (36)
390 (13)
Tumor Size (p: 0.50312)
<= 3 cm
123 (68)
1984 (64)
>3-5 cm
36 (20)
733 (24)
>5-7 cm
17 (9)
255 (8)
>7 cm
6 (3)
143 (5)
8th Clinical T (p: <0.001)
Tx
0 (0)
2 (0)
T0
14 (8)
184 (6)
Tis
1 (1)
0 (0)
T1a(mi)
0 (0)
1 (0)
T1b
25 (14)
865 (28)
T1c
24 (13)
621 (20)
T2a
27 (15)
521 (17)
T2b
18 (10)
201 (6)
T3
39 (22)
321 (10)
T4
25 (14)
228 (7)
T1a
5 (3)
156 (5)
Insufficient Records
3 (2)
11 (0)
8th Clinical N (p: <0.001)
Nx
0 (0)
1 (0)
N0
130 (71)
2709 (87)
N1
13 (7)
196 (6)
N2
35 (19)
176 (6)
N3
1 (1)
20 (1)
Insufficient
3 (2)
9 (0)
8th Clinical M (p: <0.001)
M0
131 (73)
2525 (87)
M1a
9 (5)
116 (4)
M1b
30 (17)
208 (7)
M1c
9 (5)
61 (2)
8th Clinical Stage (p: <0.001)
Occult Carcinoma
0 (0)
2 (0)
Stage 0
13 (7)
167 (5)
Stage IA1
2 (1)
141 (5)
Stage IA2
15 (8)
807 (26)
Stage IA3
12 (7)
537 (17)
Stage IB
21 (12)
424 (14)
Stage IIA
10 (5)
160 (5)
Stage IIB
29 (16)
373 (12)
Stage IIIA
41 (23)
364 (12)
Stage IIIB
15 (8)
63 (2)
Stage IVA
0 (0)
3 (0)
Stage Unknown
21 (12)
59 (2)
Extent of resection (p: <0.001)
Pneumonectomy
24 (13)
174 (6)
Bilobectomy
13 (7)
145 (5)
Lobectomy (+/-wedge)
131 (72)
2338 (75)
Segmentectomy(+/-wedge)
4 (2)
146 (5)
Wedge
10 (5)
307 (10)
Surgical Technique (p: 0.1280)
Open
124 (68)
1926 (62)
RATS
39 (21)
701 (23)
VATS
19 (10)
484 (16)
Margin Status (p: <0.001)
Positive
15 (8)
142 (5)
Negative
152 (84)
2880 (93)
Not Reported
15 (8)
88 (3)
Peri- and Post-Operative Characteristics
Surgery duration (in minutes, med, IQR) (p: 0.0172)
156 (109, 221)
135 (97, 186)
Estimated blood loss (CCs, med, IQR) (p: <0.001)
250 (100, 500)
150 (100, 300)
Duration of chest tube (in days, med, IQR) (p: 0.1364)
4 (2, 6.5)
4 (3, 7)
ICU duration (in days, med, IQR) (p: 0.0282)
2 (1, 3)
1 (1, 3)
Hospital duration (in days, med, IQR) (p: 0.7868)
6 (4, 9)
6 (4, 9)
Rate of blood transfusions (p: <0.001)
38 (21)
215 (7)
Rate of cardiac arrhythmias (p: 0.8513)
27 (15)
478 (15)
Rate of any post-op complications (p: 0.1323)
102 (56)
1567 (50)
Rate of ICU re-admittance prior to discharge (p: 0.0560)
14 (8)
143 (5)
Rate of hospital re-admittance within 30 days (p: 0.1285)
29 (17)
378 (13)
Clinical to pathologic T-Category Migration (<0.001)
Down stage
76 (42)
742 (24)
No change
40 (22)
1019 (33)
Up stage
66 (36)
1354 (43)
Unknown
0 (0)
0 (0)
Clinical to pathologic N Category Change (p: <0.001)
Down stage
48 (26)
566 (18)
No change
112 (62)
2122 (68)
Up stage
22 (12)
422 (14)
Unknown
0 (0)
1 (0)
Clinical to pathologic aggregate stage Migration (p: <0.001)
Down stage
89 (49)
879 (28)
No change
49 (27)
953 (31)
Up stage
41 (23)
1268 (41)
Unknown
3 (2)
11 (0)
Postoperative Mortality Rates
30 Day (p: 0.5109)
11 (6)
154 (5)
60 Day (p: 0.4222)
16 (9)
224 (7)
90 Day (p: 0.2216)
22 (12)
291 (9)
120 Day (p: 0.1850)
26 (14)
345 (11)
ϯ CT and PET/CT scan are after neo-adjuvant therapy for neoadjuvant patients.
Table 2. Survial impact of downstaging among neo-adjuvant patients. Prediction Variables
Hazard Ratio
95% Confidence Interval
P-value
T-category
Up-staged vs Down-staged
1.69
0.955, 2.977
0.0715
No change vs Down-staged
1.64
0.971, 2.4757
0.0642
N Category (among all patients)
Up-staged vs Down-staged
0.73
0.45, 1.20
0.2191
No change vs Down-staged
1.26
0.62, 2.58
0.5270
N Category (among clinical N2 disease patients)
Up-staged vs Down-staged
NA
NA
NA
No change vs Down-staged
1.44
0.47, 4.43
0.5261
NA – not applicable due to small sample size
8eea62084ca7e541d918e823422bd82e Conclusion
Neoadjuvant therapy was safe, but had no survival impact in this cohort, despite increased down-staging, possibly because of an inexplicably high proportion of stage I patients.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P2.16 - Treatment of Early Stage/Localized Disease (Not CME Accredited Session) (ID 965)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 2
- Moderators:
- Coordinates: 9/25/2018, 16:45 - 18:00, Exhibit Hall
-
+
P2.16-32 - Survival Implications and Factors Associated with the Anatomic Level of Incomplete Non-Small-Cell Lung Cancer (NSCLC) Resection (ID 13589)
16:45 - 18:00 | Author(s): Nicholas R. Faris
- Abstract
Background
Incomplete resection impairs NSCLC survival, but the relative significance of specific anatomic levels of margin involvement is uncertain. We examined the survival implications of the anatomic level of margin positivity.
a9ded1e5ce5d75814730bb4caaf49419 Method
We analyzed curative-intent primary NSCLC resections from 11 hospitals in 4 contiguous Dartmouth Hospital Referral Regions in West Tennessee, East Arkansas, and North Mississippi from 2009-2018. Overall survival (OS) was evaluated with Kaplan-Meier estimates and hazard ratios (HR) from Cox models.
4c3880bb027f159e801041b1021e88e8 Result
Of 3097 resections, 137 (4.4%) had positive margins. The anatomic sites of margin positivity were: mainstem bronchus 28%; peribronchial margin 24%; chest wall margin 21%; lung tissue margin 12%; great vessels 1%; mediastinum 2%; bronchial carcinoma in-situ 1%; not reported 11%.
Compared to patients with negative margins, positive margins were more likely in patients who were male (66% v 51%, p=0.0003), had poorly/undifferentiated tumor grade (45% v 34%, p=0.0143), squamous NSCLC (46% v 33%, p=0.0111), or multi-lobar tumors (15% v 5%, p<.0001). Higher pT (p<.0001) and pN (p<.0001), larger tumor size (4.2cm v 3.0cm, p<.0001), a pneumonectomy (16% v 6%, p<.0001), and an open thoracotomy (77% v 60%, p=0.0004) were also associated with margin positive resections.
5-year OS varied by the anatomic site of the margin: mainstem bronchus 35%; peribronchial margin 44%; chest wall margin 22%; lung tissue margin 23%. On unadjusted regression, positive margins at mainstem bronchial (HR=1.98, p=0.0041), chest wall (HR=2.82, p<.0001), or lung tissue (HR=2.92, p=0.0007) had a negative prognostic impact. Positive margins at chest wall (aHR=1.66, 0.0310) and lung tissue margins (aHR=2.24, p=0.0137) had the most significant prognostic impact after controlling for age, sex, race, histology, primary site, tumor grade, cT, cN, and adjuvant chemo/radiation (Table).
8eea62084ca7e541d918e823422bd82e Conclusion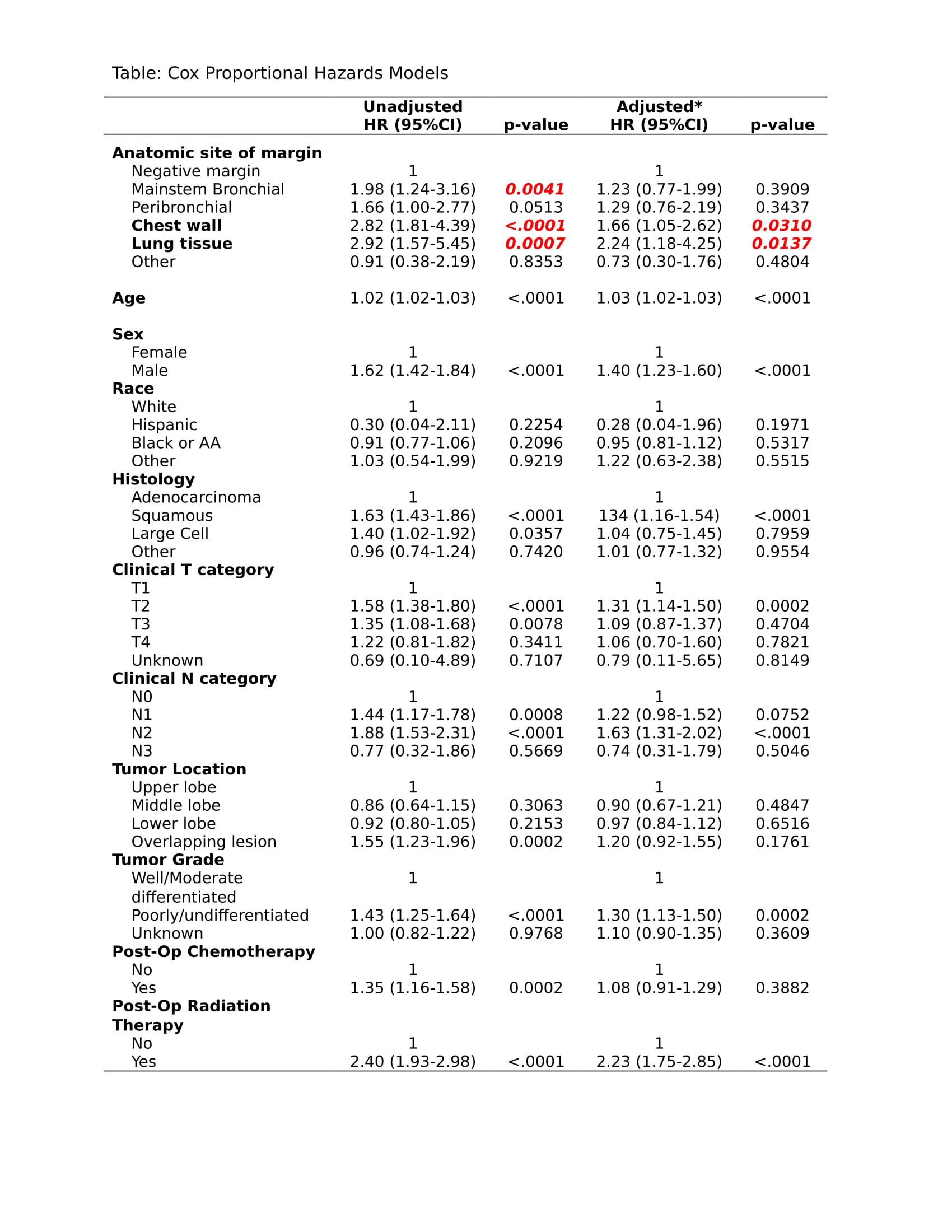
Incomplete NSCLC resection at the chest wall or the lung tissue margins connotes worse survival than other anatomic sites of margin involvement.
6f8b794f3246b0c1e1780bb4d4d5dc53 -
+
P2.16-38 - Characteristics of Preoperative N Staging in Patients with Optimal Pathologic N Staging of Non Small Cell Lung Cancer(NSCLC) (ID 14345)
16:45 - 18:00 | Author(s): Nicholas R. Faris
- Abstract
Background
Despite its prognostic value, the thoroughness of clinical and pathologic nodal staging varies significantly. We compared anatomic preoperative nodal sampling details to anatomic surgical lymphadenectomy details in resections meeting National Comprehensive Cancer Network (NCCN) quality criteria.
a9ded1e5ce5d75814730bb4caaf49419 Method
Patients had curative-intent NSCLC resections between 2009-2018 in 12 hospitals in 4 contiguous Dartmouth Hospital Referral Regions in the mid-Southern USA. Univariate statistics were calculated to summarize characteristics.
4c3880bb027f159e801041b1021e88e8 Result
1,346 of 3,297 resections (40.8%), met all 4 NCCN criteria (anatomic resection, negative margins, examination of >0 N1 nodes and ≥3 mediastinal stations). Of these 1,346, 1,114 (83%) received PET/CT scans and 148 (11%) received invasive nodal staging (all had EBUS/EUS/TBNA; 10 [16%] also had mediastinoscopy). Among 148 invasively staged patients, 72% were histologically node-negative, 19% were node-positive; 34 (23%) were down-staged and 20 (13.5%) were up-staged after surgery. Stations 4 (70.3%), 7 (73.6%), and 10 (33.8) were most frequently sampled preoperatively, 4, 7 and 9 intraoperatively (Table 2). Except for station 7, there was little overlap in anatomic station examination between invasive clinical and pathologic nodal staging.
Table 1. Preoperative characteristics among those patient with NCCN quality resections. Variables
NCCN Met
N (%)
N
1346 (40.8)
PET/CT scan
1114 (83)
Invasive staging
148 (11)
EBUS/EUS/TBNA
148 (100)
Mediastinoscopy
10 (16)
Results of invasive staging (n=148 )
Conclusive, benign
106 (72)
Conclusive, malignant
28 (19)
Inconclusive/Results not found
14 (9)
Clinical* to pathologic N category change among those receiving invasive staging (n=148)
Up-stage
20 (13.5)
No Change
94 (63.5)
Down-stage
34 (23)
*Clinical stage includes findings on PET/CT scan Table 2. Sampling information during invasive staging and surgical resections. Sampled during invasive staging
N=148 (100%)
Sampled during surgery
N=148 (100%)
Stations Resampled
N (%)
Stations not Resampled
N (%)
Stations Sampled
Station 2L
5 (3.4)
5 (3.4)
2 (1.4)
6 (4.1)
Station 2R
14 (9.5)
57 (38.5)
9 (6.1)
53 (35.8)
Station 3a
4 (2.7)
3 (2)
3 (2)
1 (0.7)
Station 3p
1 (0.7)
2 (1.4)
1 (0.7)
1 (0.7)
Station 4L
66 (44.6)
52 (35.1)
38 (25.7)
42 (28.4)
Station 4R
91 (61.5)
90 (60.8)
66 (44.6)
49 (33.1)
Station 5
7 (4.7)
59 (39.9)
7 (4.7)
52 (35.1)
Station 6
2 (1.4)
43 (29.1)
2 (1.4)
41 (27.7)
Station 7
113 (76.4)
140 (94.6)
109 (73.6)
35 (23.6)
Station 8
3 (2)
94 (63.5)
2 (1.4)
93 (62.8)
Station 9
6 (4.1)
108 (73)
5 (3.4)
104 (70.3)
Station 10R
39 (26.4)
57 (38.5)
20 (13.5)
41 (27.7)
Station 10L
24 (16.2)
81 (54.7)
30 (20.3)
60 (40.5)
N2-NOS
1 (0.7)
24 (16.2)
0 (0)
25 (16.9)
Station 11L
18 (12.2)
7 (4.7)
1 (0.7)
23 (15.5)
Station 11R
21 (14.2)
14 (9.5)
5 (3.4)
25 (16.9)
Station 12L
0 (0)
5 (3.4)
0 (0)
5 (3.4)
Station 12R
1 (0.7)
2 (1.4)
0 (0)
3 (2)
Station 13R
0 (0)
4 (2.7)
0 (0)
4 (2.7)
N1-NOS
0 (0)
103 (69.6)
0 (0)
103 (69.6)
8eea62084ca7e541d918e823422bd82e Conclusion
In a pathologic well-staged cohort of NSCLC recipients, invasive clinical nodal staging was infrequently and less-than-thoroughly used.
6f8b794f3246b0c1e1780bb4d4d5dc53


