Virtual Library
Start Your Search
Hong-Hong Yan
Author of
-
+
MA15 - Colliding Approaches - EGFR and Immunotherapy (ID 916)
- Event: WCLC 2018
- Type: Mini Oral Abstract Session
- Track: Targeted Therapy
- Presentations: 1
- Moderators:
- Coordinates: 9/25/2018, 13:30 - 15:00, Room 107
-
+
MA15.06 - Circulating Tumor DNA Portrays the Resistance Landscape to a Novel Third Generation EGFR Inhibitor, AC0010 (ID 13641)
14:05 - 14:10 | Author(s): Hong-Hong Yan
- Abstract
- Presentation
Background
In a Phase I/II dose-escalation and expansion study conducted at Guangdong Lung Cancer Institute, AC0010 demonstrated promising efficacy and good tolerability in advanced NSCLC patients with EGFR T790M-mediated resistance to previous EGFR TKIs, (NCT02330367). As disease progression (PD) with EGFR T790M-directed therapy also emerges over time, we investigated the resistance mechanisms to AC0010 in this study.
a9ded1e5ce5d75814730bb4caaf49419 Method
Serial ctDNA samples obtained from patients who developed PD with AC0010 were analyzed using ultra-deep sequencing capturing 295 cancer-related genes. Alterations that were absent before treatment and acquired at PD or that increased in abundance during treatment were identified as putative resistance mechanisms.
4c3880bb027f159e801041b1021e88e8 Result
Longitudinal plasma samples were obtained from 23 patients who progressed on AC0010 (data cut-off October 14, 2016; figure1). Putative resistance mechanisms to AC0010 were identified in 19/23 patients (>1 putative resistance mechanism was detected in some patients). EGFR amplification was the predominant resistance mechanism (21.1% [4/19 patients]), followed by TP53 loss of heterozygosity (15.8% [3/19]). EGFR C797S mutation, Met amplification and mutations in the PI3KCA pathway each occurred in 10.5% of patients (2/19). SCLC transformation, ERBB2 amplification, CD79A_A32G mutation, CDKN2A_R80 mutation, CRLF2 amplification, MLH1 amplification, Rb1 loss, and concurrent rise in the allelic fraction of tumor suppressor gene TP53 and Rb1 were each detected in 5.3% of patients (1/19). In a patient with PD following single-agent AC0010 and EGFR amplification as the putative resistance mechanism to AC0010, subsequent treatment with AC0010 plus nimotuzumab (EGFR monoclonal antibody) successfully overcame resistance, resulting in a response that lasted for 7.7 months.
8eea62084ca7e541d918e823422bd82e Conclusion
The resistance landscape to AC0010 appears to differ from that described previously with osimertinib. In this cohort of patients in China, EGFR amplification was the predominant resistance mechanism to AC0010 and could be potentially overcome by EGFR dual inhibition.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
P1.03 - Biology (Not CME Accredited Session) (ID 935)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.03-34 - Combined Molecular and Radiological Evaluation Unveils Three Subtypes of Disease Progression to a Third Generation EGFR TKI (ID 12055)
16:45 - 18:00 | Author(s): Hong-Hong Yan
- Abstract
Background
The definition of disease progression (PD) to EGFR TKIs has evolved from RECIST to a combination of clinical and RECIST evaluation. Patients with dramatic, local or gradual progression to third generation EGFR TKIs have been tailored to different subsequent treatment strategies. However, little is known about progression to third generation EGFR TKIs from molecular perspective.
a9ded1e5ce5d75814730bb4caaf49419 Method
Longitudinal plasma samples were collected from T790M-positive patients who progressed on a third generation EGFR TKI AC0010 in a phase I/II study in Guangdong Lung Cancer Institute. A pre-defined and unified molecular and radiological evaluation of PD were performed. Ultra-deep sequencing capturing 295 cancer-related genes was performed to track the changes in ctDNA to depict molecular PD, which was defined by acquired SNV/SCNV, or ≥20% increase in allelic fraction/copy number of pre-existing SNV /SCNV or both. Radiological PD was defined by RECIST.
4c3880bb027f159e801041b1021e88e8 Result
As of October 2016, 102 serial plasma samples from 23 patients with clinical PD were included. Three subtypes of PD to AC0010 were revealed (Fig1). Molecular PD occurred prior to radiological PD in 43.5% of patients (10/23), with an average lead time of 3.0 months. Molecular PD occurred concurrently with radiological PD in 39.1% of patients (9/23). Interestingly, 17.4% of patients (4/23) experienced radiological PD prior to molecular PD, with molecular PD occurred during AC0010 continuation beyond progression (CBPD) in 3 patients. Of patients experienced clinical stable PD in extracranial lesions, radiological PD occurring prior to molecular PD group (n=2) demonstrated longer duration of AC0010 CBPD than molecular PD occurring prior to (n=3) or concurrently with radiological PD groups (n=4) (Median, 5.6 months vs. 1.9 months vs. 1.8 months).
8eea62084ca7e541d918e823422bd82e Conclusion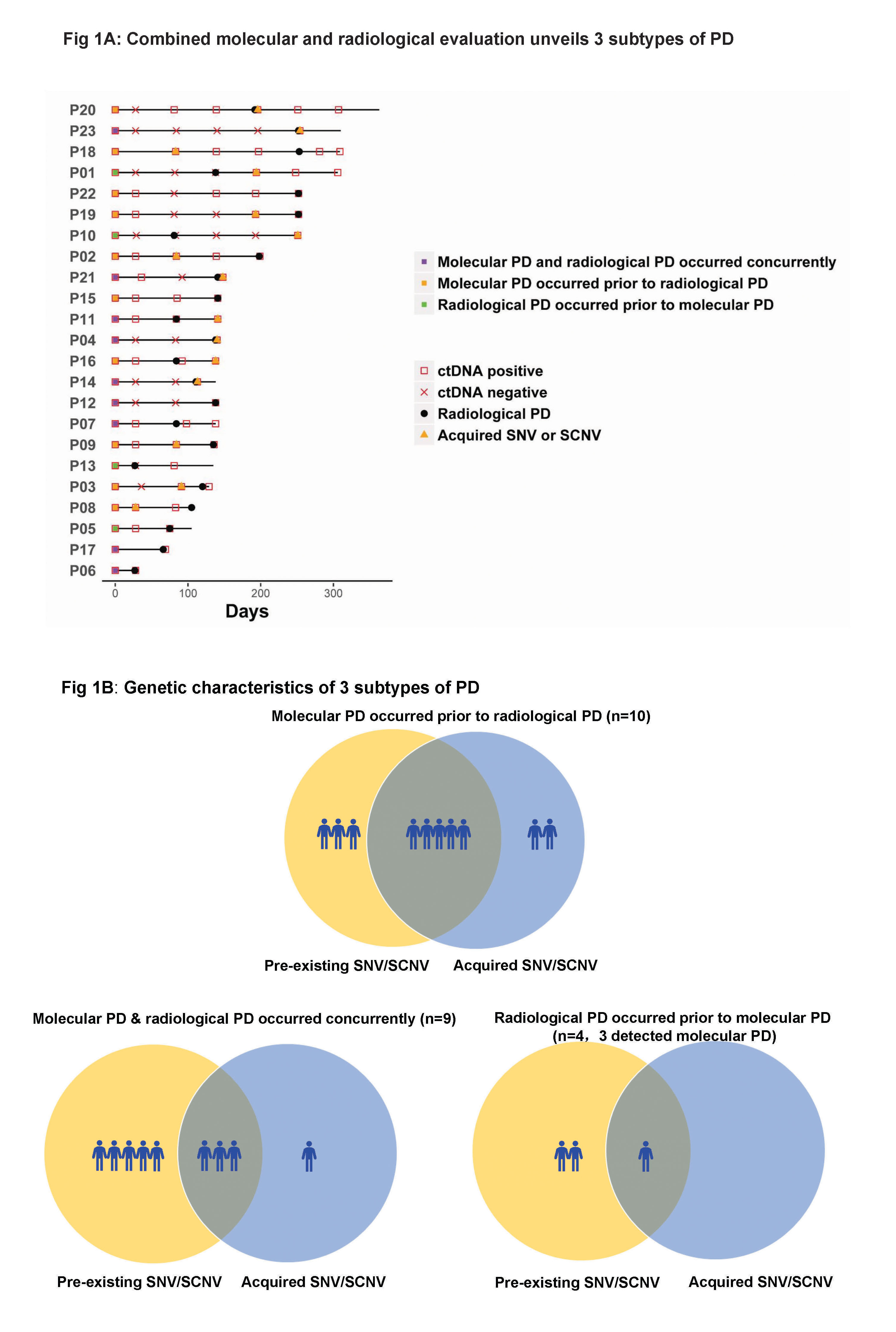
Our study revealed 3 distinct subtypes of PD to AC0010, providing insights into PD by combining molecular and radiological evaluation and might guide the optimal time for treatment switch and personalized subsequent treatments.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P3.01 - Advanced NSCLC (Not CME Accredited Session) (ID 967)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/26/2018, 12:00 - 13:30, Exhibit Hall
-
+
P3.01-64 - Preliminary Data of Diverse Therapies in Patients with Advanced Non–Small-Cell Lung Cancer Harbouring RET-Rearrangement (ID 13677)
12:00 - 13:30 | Author(s): Hong-Hong Yan
- Abstract
Background
Activating RET-rearrangement has been discovered to play a crucial role in NSCLC tumorigenesis. However, the lack of specificity narrowed efficacy of multi-kinase inhibitors (MKIs) and the optimal treatment remains unknown. In this study, we compared chemotherapy, immunotherapy and MKIs in this group of patients.
a9ded1e5ce5d75814730bb4caaf49419 Method
We retrospectively evaluated the efficacy of these three treatments in advanced, RET-rearranged NSCLC patients between January 2013 and April 2018 at our institution. RET-rearrangements were assessed by Next-generation sequencing (NGS) or any of FISH, IHC, RT-PCR. Treatment data were collected after the patients had been diagnosed with RET-rearranged advanced NSCLC. Progression-free survival (PFS) was measured from treatment start to disease progression, all-cause mortality or last follow up. Median follow-up time was 5.1months. NGS was performed to assess somatic mutation of available samples.
4c3880bb027f159e801041b1021e88e8 Result
A total of 30 patients with RET-rearrangement were investigated in this study. After the diagnosis, 15 patients, genetic profiles confirmed by NGS, received chemotherapy (n=10), checkpoint-inhibitors (n=7) and RET targeted MKI (n=6) with evaluable response. Several patients take any two of these three treatments as different line therapies. The disease control rate of chemotherapy, immunotherapy, MKI group was 70.0%, 71.43% and 50%, respectively. While the median PFS of three groups was 2.50 months, 2.70 months, 0.30 months, respectively, which of no significance. The NGS data of 10 patients showed that RET-rearrangement co-occurred with several other genes, including TP53, NTRK, CDK4, ERBB4. A low mutation burden (mean 4.5 mutations) was observed (Figure 1).
8eea62084ca7e541d918e823422bd82e Conclusion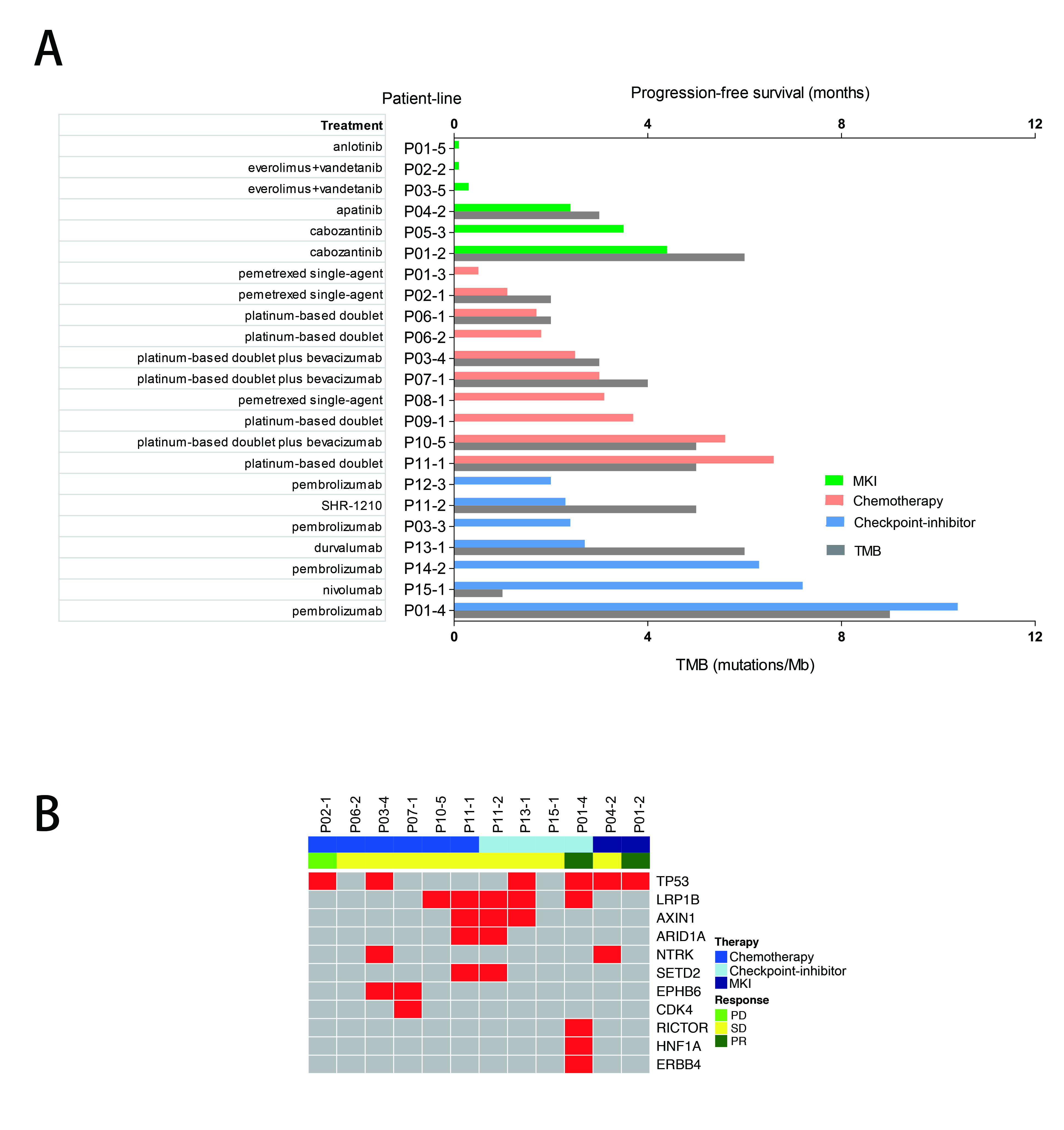
We confirmed relatively low PFS in advanced RET-rearranged NSCLC with MKIs reported in previous studies. But further investigation is warranted. Treatment with checkpoint-inhibitors seemed to encouragingly prolong PFS but a larger group of patients is needed to draw a definite conclusion.
6f8b794f3246b0c1e1780bb4d4d5dc53


