Virtual Library
Start Your Search
Jack A Jack A. Roth
Author of
-
+
MA06 - PDL1, TMB and DNA Repair (ID 903)
- Event: WCLC 2018
- Type: Mini Oral Abstract Session
- Track: Biology
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 13:30 - 15:00, Room 206 AC
-
+
MA06.02 - Prospective Immunogenomic Profiling of Non-Small Cell Lung Cancer: Genomic and Immune Profiling Updates from Project ICON (ID 13523)
13:35 - 13:40 | Author(s): Jack A Jack A. Roth
- Abstract
- Presentation
Background
Our previous work has demonstrated that higher level of genomic complexity is associated with more heterogeneous neoantigen repertoire, suppressed T cell repertoire and postsurgical relapse in localized non-small cell lung cancers (NSCLC) highlighting the complex interaction of tumor molecular and immune landscape and their impact on cancer biology and patient survival. We launched the ICON Project (Immune Genomic Profiling of NSCLC) to prospectively delineate the molecular and immune landscape of early stage NSCLC and their impact on patient survival through a multidisciplinary approach. Here we report the updated genomic and immune analyses.
a9ded1e5ce5d75814730bb4caaf49419 Method
Surgical specimens from stage I-III NSCLC were subjected to whole-exome and RNA sequencing for mutational analysis, in silico neoantigen prediction and gene expression analysis as well as T cell receptor sequencing, cytometry by time-of-flight and multiplex immunofluorescence staining.
4c3880bb027f159e801041b1021e88e8 Result
From 2016-2018, 127 patients were accrued and 50 surgical samples have undergone WES, RNAseq, TCR sequencing and immune phenotyping. Median age is 66 yrs (range: 39-86), 52% (26/50) were female and 76% (38/50) former smokers. 76% (38/50) are non-squamous carcinomas and 24% (12/50) squamous cell carcinomas. 34% have stage I disease (17/50), 30% stage II (15/50), 34% stage III (17/50) and 2% stage IV (1/50). The majority of patients had upfront surgery (45/50; 90%). With median follow-up of 19 months, 15 patients have relapsed. Median tumor mutational burden is 7.8mut/Mb and predicted neoantigen burden was 10/sample (range: 0-250). Predicted neoantigen burden is significantly correlated with tumor mutational burden (r=0.41, p=0.002). The most commonly mutated genes are TP53, KRAS, CDKN2A, PIK3CA, EGFR, BRAF, GRIN2A and ATM. C->A transversions and C->T transitions were the most common mutational subtypes. PD-1 expression and regulatory T-cell (CD4+/FoxP3+) infiltration are significantly increased in tumor tissue compared to normal tissue (p=0.003 and p=0.02 respectively), while CD3, CD8, granzyme B and CD45RO are decreased in tumor tissue compared to normal lung.
8eea62084ca7e541d918e823422bd82e Conclusion
NSCLC tumors have an immunosuppressive microenvironment compared to tumor adjacent normal lung tissues. Clinical data will be adequate to conduct genomic and immune profiling comparisons across different clinical subgroups. Mutational and neoantigen profiling are consistent with previously reported studies and correlations between molecular and immune landscapes and its impact on patient survival are ongoing.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
P1.04 - Immunooncology (Not CME Accredited Session) (ID 936)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.04-08 - Co-Residence of Patient-Derived Immune Cells in Patient-Derived Xenografts from Lung Cancer Patients (ID 11161)
16:45 - 18:00 | Author(s): Jack A Jack A. Roth
- Abstract
Background
Patient-derived xenograft (PDX) models have been shown to recapitulate many characteristics of human tumors and have been increasingly used for anticancer drug development, molecular characterization of cancer biology, and development of precision therapies. However, because PDXs are grown in immunodeficient mouse strains, they are regarded as inappropriate for preclinical evaluation of anticancer immunotherapy. Here we evaluated whether patient-derived immune cells co-exist in PDXs derived from lung cancer patients.
a9ded1e5ce5d75814730bb4caaf49419 Method
First-generation PDX (F1) was established by subcutaneously implanting human tumor tissue into non-obese diabetic-severe combined immunodeficiency (NOD-SCID) mice with a null mutation of the gene encoding for interleukin-2 receptor g (NSG). When the resulting tumors in these mice grew to about 1.5 cm in diameter, we passaged the tumors in NSG or nude mice for subsequent generations. A small piece of these PDX tissues (about 2-3 mm3) were minced into fragments and cultured in media containing human interleukin-2 (IL-2) (2000 -6000 units/ml) for up to 6 weeks. The proliferated lymphocytes for analyzed by fluorescence-activated cell sorting (FACS) with antibodies specific for human immune cell surface markers. The provenance of cultured cells was determined by DNA fingerprinting assay together with patients’ DNA samples from primary tumors and/or peripheral blood mononuclear cells (PBMC).
4c3880bb027f159e801041b1021e88e8 Result
The mean time of PDX growth in NSG mice before harvesting for culturing tumor-infiltrating lymphocytes (TILs) was 120 days (ranging from 63-292 days). TILs were successfully cultured from 8 of 25 PDXs samples (about 32%), with one from F2 PDXs and 7 from F1 PDXs. TILs from five of those PDXs were predominantly human CD3+CD8+ T cells (72% -99%), while the remaining three were predominantly human CD19+ B cells (77% - 95%). DNA fingerprint analysis showed that genotypes of TILs were identical to patients’ primary tumors and/or PBMC, demonstrating that the TILs were from the same patients as the PDXs. Further analysis showed that CD8+ T cells from PDXs were CD45RO+, with either CD62L+ or CD62L-.
8eea62084ca7e541d918e823422bd82e Conclusion
Patient-derived immune cells co-exist with PDXs in some lung cancer PDX models. Most of those immune cells were CD3+CD8+ and could be memory T cells. These results suggest that some PDXs might be used for evaluating functions of tumor resident immune cells and/or for evaluating anticancer immunotherapies.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P2.01 - Advanced NSCLC (Not CME Accredited Session) (ID 950)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/25/2018, 16:45 - 18:00, Exhibit Hall
-
+
P2.01-87 - Profiling the Symptom Burden of Patients with Metastatic NSCLC Receiving Either Chemotherapy or Targeted Therapy: Real-World Data (ID 13348)
16:45 - 18:00 | Author(s): Jack A Jack A. Roth
- Abstract
Background
An understanding of the patient experience is lacking for newly developed cancer treatments, such as targeted therapies. We profiled the patient-reported outcome (PRO)-measured symptom burden experienced by patients with metastatic non-small cell lung cancer (mNSCLC) during 6 months of conventional chemotherapy or targeted therapy.
a9ded1e5ce5d75814730bb4caaf49419 Method
During 2017, patients with mNSCLC at a single institution were recruited and completed the MD Anderson Symptom Inventory lung cancer module (MDASI-LC) at clinic visits. The MDASI-LC assesses the severity of 13 core and 3 lung-cancer-specific symptoms and 6 interference items on 0‒10 scales (0=no symptom or interference, 10=worst imaginable symptom or complete interference). Descriptive statistics for MDASI-LC scores over 6 months of treatment were summarized. Symptom trajectories for the chemotherapy patients versus the targeted-therapy patients were compared via linear mixed-effects models.
4c3880bb027f159e801041b1021e88e8 Result
Of 65 patients receiving chemotherapy and 27 receiving targeted therapy, the targeted-therapy group had more women (74% vs. 49%, P=0.029) and younger patients (57.6±12.2 vs. 64.2±9.9 years, P=0.012). Before treatment, both groups reported similar symptom burden, although sadness was worse in the targeted-therapy group (2.4±1.6 vs. 0.8±1.5, P=0.021). During the first 60 days of treatment, patients receiving chemotherapy reported significant increase in pain (estimate (est)=0.03, P=0.037) and interference with walking (est=0.04, P=0.025). Compared with those receiving chemotherapy, patients receiving targeted therapy experienced significantly less severe pain (est=‒1.17, P=0.024), fatigue (est=‒1.16, P=0.019), and shortness of breath (est=‒1.23, P=0.028) and less interference with walking (est=‒1.23, P=0.042) (figure 1). More severe dry mouth was reported by patients undergoing targeted therapy (est=1.17, P=0.027).
8eea62084ca7e541d918e823422bd82e Conclusion
This real-world data demonstrates that, compared with conventional chemotherapy, targeted therapy correlates with less impairment of physiological condition and functioning in patients with mNSCLC. Additional follow up will confirm and expand these findings about the patient experience relative to treatment response.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P3.01 - Advanced NSCLC (Not CME Accredited Session) (ID 967)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 2
- Moderators:
- Coordinates: 9/26/2018, 12:00 - 13:30, Exhibit Hall
-
+
P3.01-109 - Real-World Patient-Reported Outcome Assessment of Patients with Metastatic Non-Small Cell Lung Cancer (ID 12213)
12:00 - 13:30 | Author(s): Jack A Jack A. Roth
- Abstract
Background
Patient-Reported Outcomes (PROs) provide information on patient treatment experience. We have established a real-world Advanced Non-Small Cell Lung Holistic Registry (ANCHoR) to understand how the advent of immunotherapy impacts treatment choice, clinical outcomes, and PROs of metastatic non-small cell lung cancer (mNSCLC). The aim of this analysis is to report early results of baseline symptom status and quality of life among mNSCLC patients using the MD Anderson Symptom Inventory lung cancer module (MDASI-LC) and EuroQol-5D 5-level version (EQ-5D-5L).
a9ded1e5ce5d75814730bb4caaf49419 Method
During 2017, patients with mNSCLC at a single institution were enrolled in ANCHoR and completed the PRO questionnaires at clinic visits. MDASI-LC consists of thirteen core and three lung cancer-specific symptom severity questions, and six interference items rated on 0-10 scales (0 = no symptom or interference, 10 = worst imaginable symptom or complete interference). EQ-5D-5L captures five health state dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression rated on a five-level scale (1= no problems, 5= extreme problems). A single visual analogue scale (VAS) on EQ-5D-5L records patient self-rated health between ”best imaginable” (100) and “worst imaginable” (0) health state. Descriptive statistics for PRO scores at baseline are summarized.
4c3880bb027f159e801041b1021e88e8 Result
Forty-two patients completed baseline PROs before the start of therapy. Mean patient age was 63 years and 45% were males. For MDASI-LC, the mean scores for the core symptom, lung cancer-specific symptom, and interference subscales at baseline were 2.2 (standard deviation [SD] = 2.80), 2.1 (SD = 2.80), and 2.8 (SD = 3.10), respectively. Fatigue was the most severe symptom reported at baseline (mean = 4.1, SD = 3.01), followed by shortness of breath (mean = 3.2, SD = 2.81) and pain (mean = 3.19, SD = 3.00). The highest percentages of patients reporting moderate to severe symptom levels (score of ≥5) were 38% for fatigue, 33% for pain, 31% for drowsiness, 29% for shortness of breath and disturbed sleep, and 26% coughing. For EQ-5D-5L, 91% of patient reported problems with self-care, 81% with mobility, 48% with usual activity and anxiety, and 33% with pain. Mean EQ-5D VAS was 73.9 (SD = 18.2).
8eea62084ca7e541d918e823422bd82e Conclusion
Prior to the start of treatment, fatigue, pain, drowsiness, disturbed sleep, and coughing were the most common symptoms with fatigue, shortness of breath, and pain being the most severe. Additional follow up will confirm and expand these findings and will also allow us to examine change in PROs after first-line treatment is administered.
6f8b794f3246b0c1e1780bb4d4d5dc53 -
+
P3.01-91 - Computing the Impact of Immunotherapy on the Non-Small Cell Lung Cancer (NSCLC) Therapeutic Landscape (ID 12209)
12:00 - 13:30 | Author(s): Jack A Jack A. Roth
- Abstract
Background
The Advanced Non-Small Lung Holistic Registry (ANCHoR) is established to examine the real-world impact of immunotherapy on choice of treatment, clinical outcomes, and patient reported outcomes of patients with Stage IV NSCLC.
a9ded1e5ce5d75814730bb4caaf49419 Method
Stage IV NSCLC patients diagnosed or initiating treatment at MD Anderson from January 1, 2017 are enrolled in the ongoing ANCHoR study. Their demographic, clinicopathological, molecular, and treatment data were populated in a prospective database. Treatment patterns by line and PD-L1 status were summarized in this interim analysis.
4c3880bb027f159e801041b1021e88e8 Result
At the time of data cut off (Dec 31, 2017) 182 patients were enrolled in the registry, of which 150 were tested for PD-L1. Number of patients initiating first-, second-, and third-line treatment were 163, 42 and 7, respectively. Of the 30 patients not tested for PD-L1, 10 did not have enough tissue and 8 had actionable mutations.
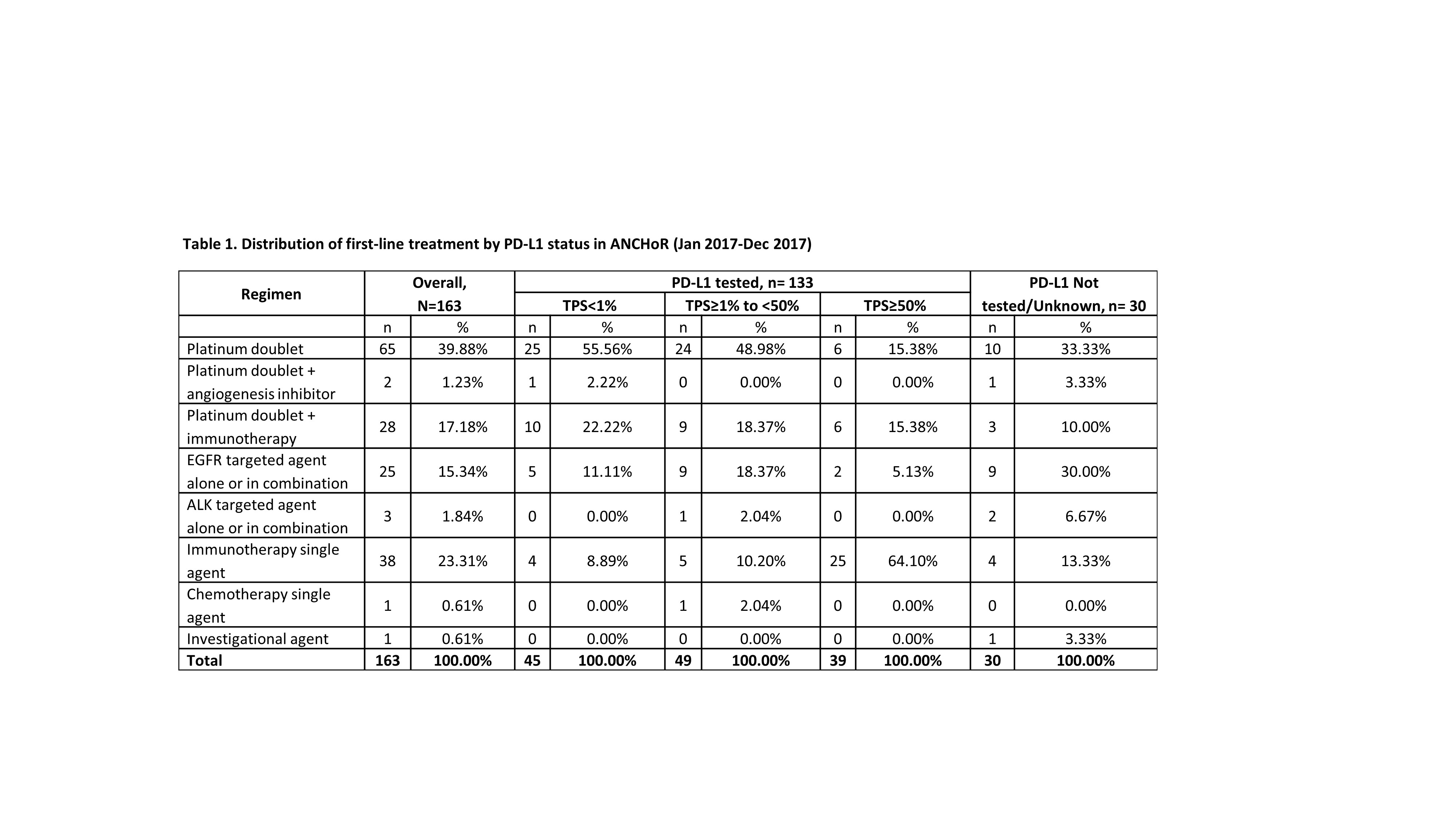
8eea62084ca7e541d918e823422bd82e Conclusion
The emergence of immunotherapy has had a dramatic impact on the first-line treatment of patient with advanced NSCLC. As of December, 2017 up to 41% of patient received immunotherapy either singly (23%) or in combination with chemotherapy. Only 40% of the patients now receive chemotherapy alone. There has been dramatic decrease in the use of chemotherapy with an anti-angiogenesis agent (1.23%). In our dataset 16% of the patients were eligible for targeted therapy as initial treatment.
6f8b794f3246b0c1e1780bb4d4d5dc53


