Virtual Library
Start Your Search
Yu-Sheng Lee
Author of
-
+
P1.17 - Treatment of Locoregional Disease - NSCLC (Not CME Accredited Session) (ID 949)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.17-11 - Pattern and Survival Impact of Neoadjuvant Treatment of Non-Small Cell Lung Cancer (NSCLC) in a Prospective Lung Resection Cohort (ID 14169)
16:45 - 18:00 | Author(s): Yu-Sheng Lee
- Abstract
Background
Neoadjuvant therapy may benefit locally-advanced NSCLC patients. We evaluated patterns of neoadjuvant therapy and the impact on stage-shift and survival.
a9ded1e5ce5d75814730bb4caaf49419 Method
All curative-intent NSCLC resections were collected from 12 hospitals in 4 contiguous Dartmouth Hospital Referral Regions in mid-Southern USA from 2009-2018. Comparisons made using Chi-square tests and non-parametric t-tests, survival impact assessed using Cox-proportional hazard models.
4c3880bb027f159e801041b1021e88e8 Result
182 of 3,297 resections (5.5%) had neoadjuvant therapy: 118 (64.8%) chemoradiation, 47 (25.8%) chemotherapy, and 17 (9.3%) radiation. Neoadjuvantly treated patients were younger, more likely to be commercially insured, have immediate preoperative brain MRI, and invasive mediastinal staging than those with primary resection (p<0.001 for all, Table 1). They also had more advanced stage, but 27% were clinical stage IA/IB.
Despite evidence of more difficult surgery, perioperative complications, hospital length of stay and postoperative mortality rates were similar to the primary resection cohort. Despite delay to surgery, they had significantly greater down-staging (p<0.001). However, down-staging had no impact on survival, regardless of type of neoadjuvant therapy (Table 2).
Table 1. Demographic and clinical characteristics between patients who received neoadjuvant therapy and those who did not. Demographic Variables
Neoadjuvant Therapy
Primary Resection Only
N
N=182
N=3115
Race (p: 0.0324)
Caucasian
125 (69)
2425 (78)
Black or AA
56 (31)
651 (21)
Asian
0 (0)
16 (1)
AI/AN
0 (0)
2 (0)
Other/NR
1 (1)
19 (1)
Age (p: <0.001)
63 (56, 69)
68 (61, 74)
Sex (p: 0.4323)
Male
104 (57)
1686 (54)
Female
78 (43)
1427 (46)
Insurance (p: <0.001)
Medicare
45 (25)
1417 (45)
Medicaid
42 (23)
438 (14)
Commercial
91 (50)
1148 (37)
Self/ None
4 (2)
112 (4)
Non-Invasive Staging
CT Scan ϯ
135 (74)
2879 (92)
PET/CT ϯ
106 (58[RO2] )
2553 (82)
Brain Scan (p: <0.001)
98 (54)
877 (28)
Invasive staging tests (p: <0.001)
No
144 (79)
2778 (89)
Yes
38 (21)
337 (11)
Histology (p: <0.001)
Adenocarcinoma
65 (40)
1547 (54)
Squamous
57 (35)
985 (34)
Other including but limited to Adenosqamous, large cell, carcinomas, and other
40 (22)
339 (11)
Grade (p: <0.001)
Well/Moderately
50 (30)
1684 (54)
Poorly/Undifferentiated
60 (33)
1040 (34)
Not Reported
66 (36)
390 (13)
Tumor Size (p: 0.50312)
<= 3 cm
123 (68)
1984 (64)
>3-5 cm
36 (20)
733 (24)
>5-7 cm
17 (9)
255 (8)
>7 cm
6 (3)
143 (5)
8th Clinical T (p: <0.001)
Tx
0 (0)
2 (0)
T0
14 (8)
184 (6)
Tis
1 (1)
0 (0)
T1a(mi)
0 (0)
1 (0)
T1b
25 (14)
865 (28)
T1c
24 (13)
621 (20)
T2a
27 (15)
521 (17)
T2b
18 (10)
201 (6)
T3
39 (22)
321 (10)
T4
25 (14)
228 (7)
T1a
5 (3)
156 (5)
Insufficient Records
3 (2)
11 (0)
8th Clinical N (p: <0.001)
Nx
0 (0)
1 (0)
N0
130 (71)
2709 (87)
N1
13 (7)
196 (6)
N2
35 (19)
176 (6)
N3
1 (1)
20 (1)
Insufficient
3 (2)
9 (0)
8th Clinical M (p: <0.001)
M0
131 (73)
2525 (87)
M1a
9 (5)
116 (4)
M1b
30 (17)
208 (7)
M1c
9 (5)
61 (2)
8th Clinical Stage (p: <0.001)
Occult Carcinoma
0 (0)
2 (0)
Stage 0
13 (7)
167 (5)
Stage IA1
2 (1)
141 (5)
Stage IA2
15 (8)
807 (26)
Stage IA3
12 (7)
537 (17)
Stage IB
21 (12)
424 (14)
Stage IIA
10 (5)
160 (5)
Stage IIB
29 (16)
373 (12)
Stage IIIA
41 (23)
364 (12)
Stage IIIB
15 (8)
63 (2)
Stage IVA
0 (0)
3 (0)
Stage Unknown
21 (12)
59 (2)
Extent of resection (p: <0.001)
Pneumonectomy
24 (13)
174 (6)
Bilobectomy
13 (7)
145 (5)
Lobectomy (+/-wedge)
131 (72)
2338 (75)
Segmentectomy(+/-wedge)
4 (2)
146 (5)
Wedge
10 (5)
307 (10)
Surgical Technique (p: 0.1280)
Open
124 (68)
1926 (62)
RATS
39 (21)
701 (23)
VATS
19 (10)
484 (16)
Margin Status (p: <0.001)
Positive
15 (8)
142 (5)
Negative
152 (84)
2880 (93)
Not Reported
15 (8)
88 (3)
Peri- and Post-Operative Characteristics
Surgery duration (in minutes, med, IQR) (p: 0.0172)
156 (109, 221)
135 (97, 186)
Estimated blood loss (CCs, med, IQR) (p: <0.001)
250 (100, 500)
150 (100, 300)
Duration of chest tube (in days, med, IQR) (p: 0.1364)
4 (2, 6.5)
4 (3, 7)
ICU duration (in days, med, IQR) (p: 0.0282)
2 (1, 3)
1 (1, 3)
Hospital duration (in days, med, IQR) (p: 0.7868)
6 (4, 9)
6 (4, 9)
Rate of blood transfusions (p: <0.001)
38 (21)
215 (7)
Rate of cardiac arrhythmias (p: 0.8513)
27 (15)
478 (15)
Rate of any post-op complications (p: 0.1323)
102 (56)
1567 (50)
Rate of ICU re-admittance prior to discharge (p: 0.0560)
14 (8)
143 (5)
Rate of hospital re-admittance within 30 days (p: 0.1285)
29 (17)
378 (13)
Clinical to pathologic T-Category Migration (<0.001)
Down stage
76 (42)
742 (24)
No change
40 (22)
1019 (33)
Up stage
66 (36)
1354 (43)
Unknown
0 (0)
0 (0)
Clinical to pathologic N Category Change (p: <0.001)
Down stage
48 (26)
566 (18)
No change
112 (62)
2122 (68)
Up stage
22 (12)
422 (14)
Unknown
0 (0)
1 (0)
Clinical to pathologic aggregate stage Migration (p: <0.001)
Down stage
89 (49)
879 (28)
No change
49 (27)
953 (31)
Up stage
41 (23)
1268 (41)
Unknown
3 (2)
11 (0)
Postoperative Mortality Rates
30 Day (p: 0.5109)
11 (6)
154 (5)
60 Day (p: 0.4222)
16 (9)
224 (7)
90 Day (p: 0.2216)
22 (12)
291 (9)
120 Day (p: 0.1850)
26 (14)
345 (11)
ϯ CT and PET/CT scan are after neo-adjuvant therapy for neoadjuvant patients.
Table 2. Survial impact of downstaging among neo-adjuvant patients. Prediction Variables
Hazard Ratio
95% Confidence Interval
P-value
T-category
Up-staged vs Down-staged
1.69
0.955, 2.977
0.0715
No change vs Down-staged
1.64
0.971, 2.4757
0.0642
N Category (among all patients)
Up-staged vs Down-staged
0.73
0.45, 1.20
0.2191
No change vs Down-staged
1.26
0.62, 2.58
0.5270
N Category (among clinical N2 disease patients)
Up-staged vs Down-staged
NA
NA
NA
No change vs Down-staged
1.44
0.47, 4.43
0.5261
NA – not applicable due to small sample size
8eea62084ca7e541d918e823422bd82e Conclusion
Neoadjuvant therapy was safe, but had no survival impact in this cohort, despite increased down-staging, possibly because of an inexplicably high proportion of stage I patients.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P2.16 - Treatment of Early Stage/Localized Disease (Not CME Accredited Session) (ID 965)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 2
- Moderators:
- Coordinates: 9/25/2018, 16:45 - 18:00, Exhibit Hall
-
+
P2.16-32 - Survival Implications and Factors Associated with the Anatomic Level of Incomplete Non-Small-Cell Lung Cancer (NSCLC) Resection (ID 13589)
16:45 - 18:00 | Presenting Author(s): Yu-Sheng Lee
- Abstract
Background
Incomplete resection impairs NSCLC survival, but the relative significance of specific anatomic levels of margin involvement is uncertain. We examined the survival implications of the anatomic level of margin positivity.
a9ded1e5ce5d75814730bb4caaf49419 Method
We analyzed curative-intent primary NSCLC resections from 11 hospitals in 4 contiguous Dartmouth Hospital Referral Regions in West Tennessee, East Arkansas, and North Mississippi from 2009-2018. Overall survival (OS) was evaluated with Kaplan-Meier estimates and hazard ratios (HR) from Cox models.
4c3880bb027f159e801041b1021e88e8 Result
Of 3097 resections, 137 (4.4%) had positive margins. The anatomic sites of margin positivity were: mainstem bronchus 28%; peribronchial margin 24%; chest wall margin 21%; lung tissue margin 12%; great vessels 1%; mediastinum 2%; bronchial carcinoma in-situ 1%; not reported 11%.
Compared to patients with negative margins, positive margins were more likely in patients who were male (66% v 51%, p=0.0003), had poorly/undifferentiated tumor grade (45% v 34%, p=0.0143), squamous NSCLC (46% v 33%, p=0.0111), or multi-lobar tumors (15% v 5%, p<.0001). Higher pT (p<.0001) and pN (p<.0001), larger tumor size (4.2cm v 3.0cm, p<.0001), a pneumonectomy (16% v 6%, p<.0001), and an open thoracotomy (77% v 60%, p=0.0004) were also associated with margin positive resections.
5-year OS varied by the anatomic site of the margin: mainstem bronchus 35%; peribronchial margin 44%; chest wall margin 22%; lung tissue margin 23%. On unadjusted regression, positive margins at mainstem bronchial (HR=1.98, p=0.0041), chest wall (HR=2.82, p<.0001), or lung tissue (HR=2.92, p=0.0007) had a negative prognostic impact. Positive margins at chest wall (aHR=1.66, 0.0310) and lung tissue margins (aHR=2.24, p=0.0137) had the most significant prognostic impact after controlling for age, sex, race, histology, primary site, tumor grade, cT, cN, and adjuvant chemo/radiation (Table).
8eea62084ca7e541d918e823422bd82e Conclusion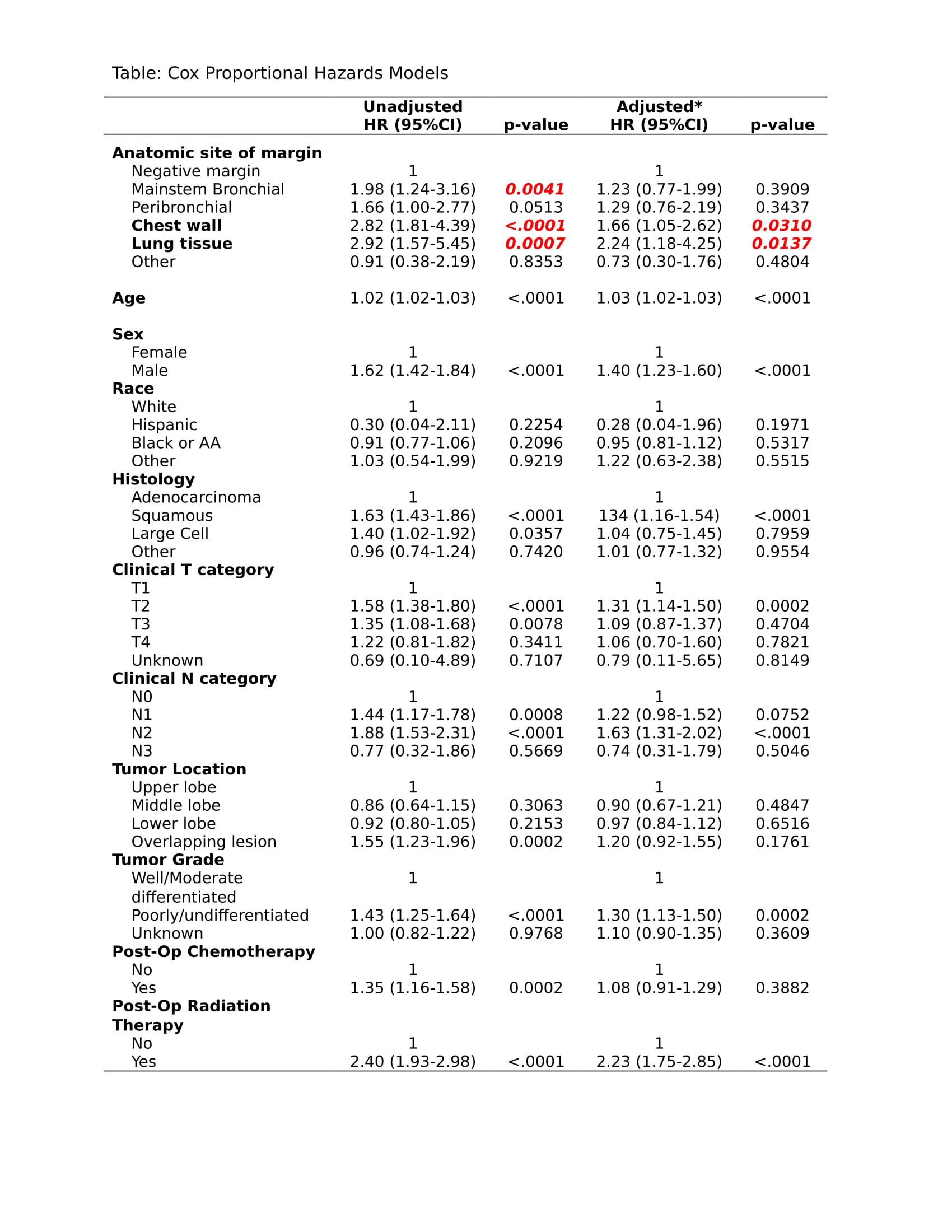
Incomplete NSCLC resection at the chest wall or the lung tissue margins connotes worse survival than other anatomic sites of margin involvement.
6f8b794f3246b0c1e1780bb4d4d5dc53 -
+
P2.16-38 - Characteristics of Preoperative N Staging in Patients with Optimal Pathologic N Staging of Non Small Cell Lung Cancer(NSCLC) (ID 14345)
16:45 - 18:00 | Author(s): Yu-Sheng Lee
- Abstract
Background
Despite its prognostic value, the thoroughness of clinical and pathologic nodal staging varies significantly. We compared anatomic preoperative nodal sampling details to anatomic surgical lymphadenectomy details in resections meeting National Comprehensive Cancer Network (NCCN) quality criteria.
a9ded1e5ce5d75814730bb4caaf49419 Method
Patients had curative-intent NSCLC resections between 2009-2018 in 12 hospitals in 4 contiguous Dartmouth Hospital Referral Regions in the mid-Southern USA. Univariate statistics were calculated to summarize characteristics.
4c3880bb027f159e801041b1021e88e8 Result
1,346 of 3,297 resections (40.8%), met all 4 NCCN criteria (anatomic resection, negative margins, examination of >0 N1 nodes and ≥3 mediastinal stations). Of these 1,346, 1,114 (83%) received PET/CT scans and 148 (11%) received invasive nodal staging (all had EBUS/EUS/TBNA; 10 [16%] also had mediastinoscopy). Among 148 invasively staged patients, 72% were histologically node-negative, 19% were node-positive; 34 (23%) were down-staged and 20 (13.5%) were up-staged after surgery. Stations 4 (70.3%), 7 (73.6%), and 10 (33.8) were most frequently sampled preoperatively, 4, 7 and 9 intraoperatively (Table 2). Except for station 7, there was little overlap in anatomic station examination between invasive clinical and pathologic nodal staging.
Table 1. Preoperative characteristics among those patient with NCCN quality resections. Variables
NCCN Met
N (%)
N
1346 (40.8)
PET/CT scan
1114 (83)
Invasive staging
148 (11)
EBUS/EUS/TBNA
148 (100)
Mediastinoscopy
10 (16)
Results of invasive staging (n=148 )
Conclusive, benign
106 (72)
Conclusive, malignant
28 (19)
Inconclusive/Results not found
14 (9)
Clinical* to pathologic N category change among those receiving invasive staging (n=148)
Up-stage
20 (13.5)
No Change
94 (63.5)
Down-stage
34 (23)
*Clinical stage includes findings on PET/CT scan Table 2. Sampling information during invasive staging and surgical resections. Sampled during invasive staging
N=148 (100%)
Sampled during surgery
N=148 (100%)
Stations Resampled
N (%)
Stations not Resampled
N (%)
Stations Sampled
Station 2L
5 (3.4)
5 (3.4)
2 (1.4)
6 (4.1)
Station 2R
14 (9.5)
57 (38.5)
9 (6.1)
53 (35.8)
Station 3a
4 (2.7)
3 (2)
3 (2)
1 (0.7)
Station 3p
1 (0.7)
2 (1.4)
1 (0.7)
1 (0.7)
Station 4L
66 (44.6)
52 (35.1)
38 (25.7)
42 (28.4)
Station 4R
91 (61.5)
90 (60.8)
66 (44.6)
49 (33.1)
Station 5
7 (4.7)
59 (39.9)
7 (4.7)
52 (35.1)
Station 6
2 (1.4)
43 (29.1)
2 (1.4)
41 (27.7)
Station 7
113 (76.4)
140 (94.6)
109 (73.6)
35 (23.6)
Station 8
3 (2)
94 (63.5)
2 (1.4)
93 (62.8)
Station 9
6 (4.1)
108 (73)
5 (3.4)
104 (70.3)
Station 10R
39 (26.4)
57 (38.5)
20 (13.5)
41 (27.7)
Station 10L
24 (16.2)
81 (54.7)
30 (20.3)
60 (40.5)
N2-NOS
1 (0.7)
24 (16.2)
0 (0)
25 (16.9)
Station 11L
18 (12.2)
7 (4.7)
1 (0.7)
23 (15.5)
Station 11R
21 (14.2)
14 (9.5)
5 (3.4)
25 (16.9)
Station 12L
0 (0)
5 (3.4)
0 (0)
5 (3.4)
Station 12R
1 (0.7)
2 (1.4)
0 (0)
3 (2)
Station 13R
0 (0)
4 (2.7)
0 (0)
4 (2.7)
N1-NOS
0 (0)
103 (69.6)
0 (0)
103 (69.6)
8eea62084ca7e541d918e823422bd82e Conclusion
In a pathologic well-staged cohort of NSCLC recipients, invasive clinical nodal staging was infrequently and less-than-thoroughly used.
6f8b794f3246b0c1e1780bb4d4d5dc53


