Virtual Library
Start Your Search
Puyuan Xing
Author of
-
+
P1.01 - Advanced NSCLC (Not CME Accredited Session) (ID 933)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 2
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.01-101 - Survival Benefit of Sequential Therapy in ALK Positive Olioprogressive NSCLC Patients After Crizotinib Resistance (ID 13110)
16:45 - 18:00 | Author(s): Puyuan Xing
- Abstract
Background
Crizotinib is recommended as first-line standard therapy for advanced anaplastic lymphoma kinase (ALK) positive non-small cell lung cancer (NSCLC).Despite initial efficacy of crizotinib, patients ultimately acquired resistance, which leading to tumor progression within one year of crizotinib initiation. However,optimal sequential therapy option among patients with olioprogressive disease remains unknown.Therefore, this study aimed to explore which therapy pattern was advantageous for those patients with extracranial and intracranial metastasis.
a9ded1e5ce5d75814730bb4caaf49419 Method
This retrospective single-center study enrolled 116 Chinese advanced ALK positive NSCLC patients resistant to prior crizotinib treatment,between Jan 2013 and Dec 2016.Based on the site of olioprogressive metastasis, patients were divided into two groups: extracranial (n=57,49.1%) and intracranial (n=59, 50.9%).Sequential therapies mainly included continuation of crizotinib with local therapeutics and next-generation ALK inhibitors. The primary end-point was median overall survival (OS) from the time of crizotinib resistance to death or last follow-up.
4c3880bb027f159e801041b1021e88e8 Result
Among 116 patients,45 patients (38.8%) had died. For extracranial group,26 patients continued crizotinib with local treatment (local radiotherapy or ablation therapy),31 received next-generation ALK inhibitor. Patients followed by next-generation ALK inhibitor beyond crizotinib resistance exhibited a longer OS of 27.6 months than crizotinib continuation with local therapy of 10.8months(p=0.009).Among intracranial group,36 patients continued crizotinib with local treatment including whole brain radiotherapy or stereotactic radiotherapy,23 were next-generation ALK inhibitors users. Continution of crizotinib with local therapy had a non-inferior OS than next-generation ALK inhibitors (28.9 month vs 32.8months, p=0.538) .
8eea62084ca7e541d918e823422bd82e Conclusion
Next-generation ALK inhibitors had more survival benefit than continuation of crizotinib with local therapy for extracranial olioprogressive patients.While crizotinib continuation with local therapy might be a feasible strategy among patients with intracranial olioprogressive metastasis after crizotinib resistance.
6f8b794f3246b0c1e1780bb4d4d5dc53 -
+
P1.01-58 - Variants Distribution and Heterogeneity of Outcomes to Crizotinib in ALK-Rearranged Chinese Non-Small Cell Lung Cancers (ID 13940)
16:45 - 18:00 | Author(s): Puyuan Xing
- Abstract
Background
ALK-rearranged NSCLC is a unique molecular subgroup with high sensitivity to ALK inhibitors. Crizotinib, a FDA-approved tyrosine kinase inhibitor for ALK-rearranged NSCLCs, showed remarkable response in ALK-positive NSCLC. However, the magnitude and duration of clinical responses to crizotinib among different ALK variants are found to be heterogeneous, and studies about the clinical outcomes showed contradict conclusions.
a9ded1e5ce5d75814730bb4caaf49419 Method
We collected sequencing information from 110 ALK-positive Chinese NSCLC patients, whose tissue or plasma biopsies were sequenced in a CLIA-certified genomic profiling laboratory. Sequencing results were reviewed with the intent of studying ALK rearrangement distribution and clinical outcomes to crizotinib.
4c3880bb027f159e801041b1021e88e8 Result
A total of 134 ALK rearrangements were identified in this cohort, with 39 unique rearrangements. EML4 was the most common ALK rearrangement partner, with variant 3 (v3) as the most frequent variants (42.7%) of EML4-ALK fusion, accounting for 71.6% (96/134) of all the rearrangements in 87.3% (96/110) patients. For EML4-ALK positive patients after crizotinib treatment (n=96), survival analysis revealed that patients with EML4-ALK only displayed favorable PFS (10.0 vs 7.2 months, p=0.037) and OS (36.0 vs 20.0 months, p=0.037) than those combined with other fusions. In vitro data reported that variant v3 and v5 was structurally stable and less sensitive to ALK inhibitors due to the lack of TAPE domain. In this study, patients harboring v3 and v5 displayed significantly inferior OS than those with other variants (31 vs 37.6 months, p=0.010). For all the ALK-rearranged patients (n=110), no significant difference was observed between the survival of EML4-ALK and non-EML4-ALK (PFS, 9.4 vs 14.5 months, p=0.61; OS, 35.1 vs 35.5 months, p=0.58), below and above 40-years (PFS, 7.3 vs 11.3 months, p=0.23; OS, 25.4 vs 35.5 months, p=0.69).
8eea62084ca7e541d918e823422bd82e Conclusion
This study demonstrated the distribution pattern of ALK rearrangements in Chinese NSCLCs, and illustrated the clinical outcomes of ALK-positive patients in different sub-groups. We hope this study could improve basic knowledge of ALK rearrangement and might be helpful for clinicians in choosing patients for appropriate medical treatment. Moreover, these findings advocate for more comprehensive ALK genomic profiling and validation of current results of clinical outcomes in large populations.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P1.13 - Targeted Therapy (Not CME Accredited Session) (ID 945)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.13-18 - Exploring the Resistance Mechanism of Osimertinib and Monitoring the Treatment Response Using Plasma ctDNA in Chinese NSCLC Patients (ID 13297)
16:45 - 18:00 | Author(s): Puyuan Xing
- Abstract
Background
Osimertinib (AZD9291; Tagrisso) is a third generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) known to be effective for patients harboring the EGFR-T790M variant, which is accounts for more than half of the acquired resistance mechanisms to the first generation EGFR-TKIs. However, limited osimertinib resistance-mechanism was reported. Study on potential osimertinib-resistance mechanisms in advanced NSCLC is necessary.
a9ded1e5ce5d75814730bb4caaf49419 Method
This study enrolled eight T790M-positive (tissue validated) patients, treated with osimertinib after first generation EGFR-TKI (Erlotinib, Gefitinib, Icotinib) resistance and progressed rapidly. Serial plasma samples were collected until disease progressed. Plasma DNA was extracted and sequenced by target-capture deep sequencing of 1021 previously annotated genes related to solid tumors. Clonal EGFR T790M mutation was defined if mutation was in the cluster with the highest mean variated allele frequency with PyClone, and otherwise subclonal EGFR T790M mutation. Molecular tumor burden index (mTBI) was calculated with the mean variant allele frequency of mutations in trunk clonal population.
4c3880bb027f159e801041b1021e88e8 Result
The median progression-free survival (PFS) of these eight rapidly-progressed patients was 3.82 months [95% CI 2.05-5.01] .Targeted capture sequencing of pretreatment ctDNA showed all of the eight patients (100%) were EGFR-positive (Exon19del [n=6] and L858R [n=2]), and seven patients (88%) harbored EGFR T790M mutation, except for the only one patient (P006) who showed an extremely low level of ctDNA. During the Osimertinib treatment, five patients (63%) had osimertinib resistance-related mutations: EGFR C797S (in cis position), G724S, KRAS G12D, PIK3CA E542K, EGFR amplification, and ERBB2 amplification. Among them, two patients had more than one resistance mechanisms: patient P034 had EGFR G724S, KRAS G12D and EGFR amplification, simultaneously; patient P013 had amplification in both EGFR and ERBB2. Other potential resistance mechanisms were identified including EGFR T751I and K754E mutations in P002 and ERBB2 S603 in P013. Notably, the only one patient (P004) who had not been detected to have any known osimertinib resistance mechanism but progressed in 3 months, was demonstrated to harbor a subclonal EGFR T790M mutation by analysis of ctDNA clonal structure. Serial ctDNA monitoring showed mTBI increased when disease progressed in 88% (7/8) patients, except P006, whose mutation were negative at second (stable disease) and third (progressed disease) therapeutic evaluations due to the extremely low level of ctDNA.
8eea62084ca7e541d918e823422bd82e Conclusion
This study presented comprehensive the resistance mechanism of osimertinib progressed rapidly in ctDNA including multiple mechanisms co-occurred in same patient. Serial monitoring of plasma ctDNA may be a promising approach to explore resistance mechanism and monitored the treatment response of third generation EGFR-TKI.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P2.01 - Advanced NSCLC (Not CME Accredited Session) (ID 950)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/25/2018, 16:45 - 18:00, Exhibit Hall
-
+
P2.01-109 - Treatment Duration—A More Reasonable Definition to Evaluate the Efficacy of Crizotinib in ALK Positive Advanced NSCLC (ID 13216)
16:45 - 18:00 | Author(s): Puyuan Xing
- Abstract
Background
Crizotinib has demonstrated its superior efficacy in ALK positive NSCLC patients when used as first-line regimen, with a median overall survival (OS) of more than 4 years, whereas a median progression-free survival (PFS) of only 10.9 months. Patients who continued crizotinib beyond progressive disease (CBPD) could still obtain additional survival benefits of 6-8 months after disease progression. In terms of that, PFS, the frequently used primary endpoint in clinical trials, may not be able to provide accurate information on impact of this intervention in multiple lines therapy. Here we proposed “treatment duration” as an intermediate clinical endpoint between PFS and OS that further define efficacy of crizotinib in multiple lines of treatment and reported the exploratory data in a real-world cohort.
a9ded1e5ce5d75814730bb4caaf49419 Method
We retrospectively enrolled 150 ALK positive NSCLC patients who had acquired crizotinib resistance from Aug 2011 to May 2017. The median PFS of crizotinib and median OS (from crizotinib initiation) were analyzed. Treatment duration of crizotinib, the time from crizotinib initiation to discontinuation, was also calculated.
4c3880bb027f159e801041b1021e88e8 Result
The median PFS of crizotinib in the 150 ALK positive advanced NSCLC patients was 14.4 months (95% CI: 11.3-17.4). Overall, 58 patients (38%) continued CBPD and the median post-progression PFS was 10.4 months, resulting in a median treatment duration of crizotinib in the total cohort of 20.2 months (95% CI: 14.3-26.0). And median OS was 30.1months (95% CI: 21.3-38.8). 77 (51%) patients who received crizotinib as first-line therapy exhibited both longer median PFS (17.7 months, 95% CI: 12.5-22.9; vs. 12.2months, 95% CI: 7.6-16.8) and median OS (35.2 months, 95% CI: 23.2-47.1; vs. 25.4 months, 95% CI: 14.4-36.3) compared to those selected crizotinib as second- line and above therapy. The treatment duration among them were 23.1 months (95% CI: 14.7-31.4) and 18.9 months (95% CI: 11.4-26.3), respectively.
8eea62084ca7e541d918e823422bd82e Conclusion
Crizotinib showed superior efficacy in ALK positive NSCLC patients. Treatment duration may be more reasonable to define the efficacy of crizotinib in multiple lines therapy of ALK positive NSCLC.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P2.13 - Targeted Therapy (Not CME Accredited Session) (ID 962)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 2
- Moderators:
- Coordinates: 9/25/2018, 16:45 - 18:00, Exhibit Hall
-
+
P2.13-04 - Outcomes of ALK-Positive Non-Small-Cell Lung Cancer (NSCLC) Patients Treated with Crizotinib: A Multicenter Cohort Retrospective Study. (ID 13304)
16:45 - 18:00 | Author(s): Puyuan Xing
- Abstract
Background
Crizotinib has demonstrated promising efficacy in patients with anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC) in clinical trials. However, there is lack of multicenter real-world data summary with large cohort of patients, especially in China. We conducted this multicenter and retrospective study to assess the outcomes of crizotinib in, to our knowledge, the largest cohort of patients with ALK-positive advanced NSCLC.
a9ded1e5ce5d75814730bb4caaf49419 Method
We reviewed medical records of 484 unselected ALK-positive NSCLC patients treated with crizotinib at five cancer centers in China from January 2013 to November 2017. Clinical data were collected from crizotinib initiation to RECIST-defined progressive disease (PD), and post-PD systemic treatment outcomes were also analyzed.
4c3880bb027f159e801041b1021e88e8 Result
A total of 428 eligible ALK-positive NSCLC patients were enrolled. Among them, 273 (63.8%) patients received crizotinib as first-line treatment. The median progression-free survival (PFS) and overall survival (OS) from crizotinib initiation were 14.4 months (95%CI: 12.4-16.4) and 53.4 months (95% CI: 33.7-73.1) respectively. In the subgroup analysis, patients received crizotinib as first-line treatment showed a higher disease control rate (DCR) and longer median OS with statistical significance compared with second-/ further line crizotinib treatment (94.8%, not estimated vs. 89.0%, 40.5 months, respectively). For 261 patients with RECIST-defined PD, multivariate COX analysis revealed that patients who received first-line crizotinib (P=0.013), continued crizotinib beyond progressive disease (CBPD) (P=0.011) and received next-generation ALKis after crizotinib failure (P<0.001) were associated with improved survival both from crizotinib progression and from the first crizotinib dose.
8eea62084ca7e541d918e823422bd82e Conclusion
This study demonstrated the clinically meaningful benefit of crizotinib treatment in the largest cohort of Chinese ALK positive NSCLC patients. CBPD and next-generation ALK TKI treatment may provide survival improvement after RECIST-defined progression on crizotinib.
6f8b794f3246b0c1e1780bb4d4d5dc53 -
+
P2.13-05 - Real-World Clinical Benefit of Continuing Crizotinib Beyond Progression Disease (CBPD) in Patients with Advanced ALK-Positive NSCLC. (ID 13353)
16:45 - 18:00 | Author(s): Puyuan Xing
- Abstract
Background
Most ALK-positive NSCLC patients treated with crizotinib would ultimately develop progressive disease (PD), and continuing crizotinib beyond initial PD (CBPD) may be potentially beneficial. We aim to evaluate the survival outcomes of patients with crizotinib resistance in real-world setting and to explore the clinical efficacy of continuing CBPD treatment.
a9ded1e5ce5d75814730bb4caaf49419 Method
A total of 261 ALK-positive NSCLC patients treated with crizotinib experienced RECIST-defined PD and were included in this multi-center retrospective analysis. Clinicopathologic characteristics, progressive pattern, post-PD treatment and overall survival (OS) were compared between patients continuing CBPD and those not.
4c3880bb027f159e801041b1021e88e8 Result
140 patients who continued crizotinib after disease progression were allocated to CBPD group and others were non-CBPD group. Two-sided Chi-square test showed that patients who never smoked (P=0.047), with ECOG 0-1(P=0.001), isolated intracranial progression (P<0.001) and <median PFS of initial crizotinib (P=0.002) were more likely in the CBPD group. At the analysis, 84 patients had re-PD and the median duration of crizotinib treatment post-PD was 6.8 months (95%CI: 3.639-9.869). The median OS for the overall population from the time of PD (post-PD OS) was 15.3 months (95%CI: 11.376-19.181), and was significantly longer in CBPD patients than non-CBPDs (24.1 months vs. 8.5 months, 95% CI: 0.326-0.669, HR 0.467, P<0.001). Furthermore, next-generation ALK inhibitors (ALKis) following crizotinib failure was associated with improved post-PD OS (24.9 months vs. 10.7 months, 95% CI: 0.307-0.686, HR 0.459, P<0.001).
8eea62084ca7e541d918e823422bd82e Conclusion
Continuing CBPD treatment after crizotinib resistance favorably impact survival outcomes of advanced ALK-positive NSCLC patients in the real-world. Next-generation ALKis may provide survival improvement, but comparative studies between different subsequent treatment options after PD on crizotinib are still needed.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P3.01 - Advanced NSCLC (Not CME Accredited Session) (ID 967)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/26/2018, 12:00 - 13:30, Exhibit Hall
-
+
P3.01-103 - Efficacy of Crizotinib in Chinese Non-Small Cell Lung Cancer Patients with Brain Metastasis: A Multicenter Retrospective Study (ID 12578)
12:00 - 13:30 | Author(s): Puyuan Xing
- Abstract
Background
Brain metastasis in advanced non-small cell lung cancer (NSCLC) patients is often considered as a terminal stage. Crizotinib is a small-molecule tyrosine kinase inhibitor (TKI) for ALK-rearranged NSCLC patients. Herein we conducted a multi-center retrospective study to explore how crizotinib affects the control of brain metastasis and survival outcomes among advanced ALK-rearranged NSCLC patients with brain metastasis in Chinese population.
a9ded1e5ce5d75814730bb4caaf49419 Method
We reviewed medical records of 484 unselected ALK-positive NSCLC patients treated with crizotinib at five cancer centers in China from January 2013 to November 2017. Patients developing brain metastasis either before or during the crizotinib treatment were enrolled. Survival outcomes were analyzed with Kaplan-Meier method and prognostic factors were analyzed with multivariate COX analysis.
4c3880bb027f159e801041b1021e88e8 Result
A total of 174 patients were enrolled into this study, of whom 95 patients had baseline brain metastasis and 79 patients developed brain metastasis during crizotinib treatment. Among patients with baseline brain metastasis, the median intracranial progression-free survival (PFS) was 15.34 months [95% confidence interval (CI): 10.62-20.07] and median overall survival (OS) was 53.38 months (95%CI: 30.58-76.17). The intracranial objective response rate (ORR) was 17.1%, and the intracranial disease control rate (DCR) was 88.6%. Multivariate COX analysis revealed that patients receiving first-line crizotinib [>first-line vs. first-line, hazard ratio (HR): 2.44, 95%CI: 1.05-5.68, p=0.038], withtout intracranial progression during crizotinib treatment (with vs. without intracranial progression, HR: 18.68, 95%CI: 2.43-143.31, p=0.005) were associated with better OS, while age, sex, number of brain lesions, and operation/radiation therapy for brain metastasis were not significantly associated with OS. Among patients developing brain metastasis during crizotinib treatment, the median OS was 35.64 months (95%CI: not reached). Multivariate COX analysis revealed that brain progression only (brain progression only vs. both brain and extracranial progression, HR: 0.23, 95%CI: 0.08-0.71, p=0.011) was associated with better OS, while age, sex, line of crizotinib treatment, treatment after progression and operation/radiation therapy for brain metastasis were not significantly associated with OS.
8eea62084ca7e541d918e823422bd82e Conclusion
Advanced ALK-rearranged NSCLC patients with baseline brain metastasis could still benefit from crizotinib treatment. However, brain progression during crizotinib treatment may be associated with worse survival outcomes.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P3.13 - Targeted Therapy (Not CME Accredited Session) (ID 979)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/26/2018, 12:00 - 13:30, Exhibit Hall
-
+
P3.13-17 - A Retrospective Study: Central Nervous System Response to Osimertinib in Patients with Advanced NSCLC (ID 11937)
12:00 - 13:30 | Author(s): Puyuan Xing
- Abstract
Background
Central nervous system (CNS) metastases are common in patients with non-small-cell lung cancer (NSCLC). More than 30% of patients who progress during or after treatment with epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs) have CNS metastases. Osimertinib, a third-generation EGFR-TKI, has been demonstrated promising intracranial efficacy in patients with advanced NSCLC from several large scale randomized control trials. We aimed to explore clinical impact of osimertinib for patients with CNS metastases, advanced NSCLC in real world setting.
a9ded1e5ce5d75814730bb4caaf49419 Method
Patients with advanced NSCLC who received osimertinib after progression of prior EGFR-TKIs and CNS metastases on baseline brain scan were retrospectively collected from Cancer Hospital Chinese Academy of Medical Sciences. Primary outcome was objective response rate (ORR) and secondary objectives were disease control rate (DCR), progression-free survival (PFS), time to tumor response, median best percentage change from baseline in CNS target lesion (TL) size and safety.
4c3880bb027f159e801041b1021e88e8 Result
Between Apr 1, 2017, and Dec 30, 2017, 22 patients met selection criteria, 15 with ≥1 measurable CNS lesion (RECIST 1.1) were included in CNS evaluable for response (cEFR) set. The median duration of follow-up was 6.5 months. For overall 22 patients, ORR and DCR were 40.9% and 86.4%, respectively, with median PFS of 8.5 months (95% CI 4.1, 13.0). Of 15 patients in cEFR set, CNS ORR was 53.3% with complete responses reported in 3 patients (20.0%). Median best percentage change from baseline in CNS TL size was -40% (range: -100% ~ +60%) and median time to CNS tumor response was 1.3 months. CNS DCR was 80.0%. Median CNS PFS was not reached. Safety profile was acceptable and no new unexpected findings were found.
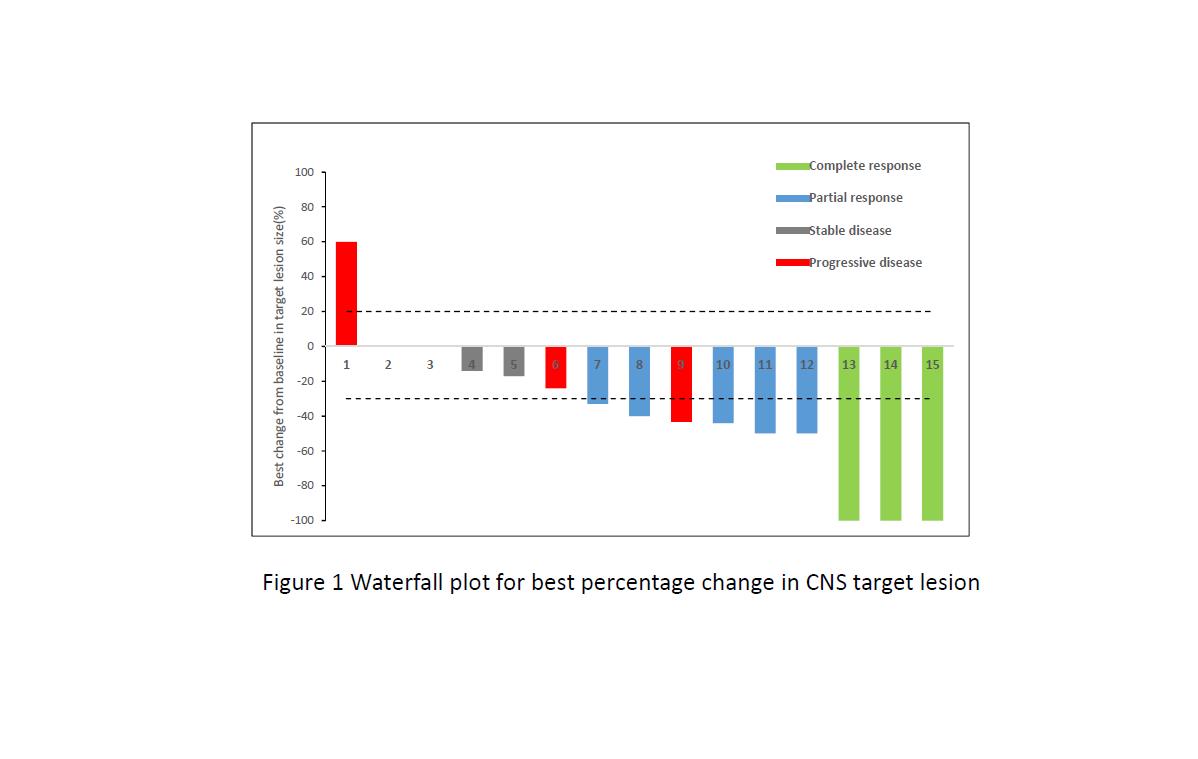
8eea62084ca7e541d918e823422bd82e Conclusion
This real world analysis further confirmed that osimertinib was indeed demonstrated clinically meaningful efficacy against CNS metastases in Chinese patients with advanced NSCLC.
6f8b794f3246b0c1e1780bb4d4d5dc53


