Virtual Library
Start Your Search
Angshu Bhowmik
Author of
-
+
MA20 - Implementation of Lung Cancer Screening (ID 923)
- Event: WCLC 2018
- Type: Mini Oral Abstract Session
- Track: Screening and Early Detection
- Presentations: 1
- Moderators:
- Coordinates: 9/25/2018, 15:15 - 16:45, Room 206 F
-
+
MA20.01 - Lung Cancer Screenee Selection by USPSTF versus PLCOm2012 Criteria – Preliminary ILST Findings (ID 14331)
15:15 - 15:20 | Author(s): Angshu Bhowmik
- Abstract
- Presentation
Background
Background
The National Lung Screening Trial showed that lung cancer screening of high-risk individuals with low dose computed tomography can reduce lung cancer mortality by 20%. Critically important is enrolling high-risk individuals. Most current guidelines including the United States Preventive Services Task Force (USPSTF) and Center for Medicare and Medicaid Services (CMS) recommend screening using variants of the NLST eligibility criteria: smoking ≥30 pack-years, smoking within 15 years, and age 55-80 and 55-77 years. Many studies indicate that using accurate risk prediction models is superior for selecting individuals for screening, but these findings are based on retrospective analyses. The International Lung Screen Trial(ILST) was implemented to prospectively identify which approach is superior.
a9ded1e5ce5d75814730bb4caaf49419 Method
Methods
ILST is a multi-centred trial enrolling 4000 participants. Individuals will be offered screening if they are USPSTF criteria positive or have PLCOm2012 model 6-year risk ≥1.5%. Participants will receive two annual screens and will be followed for six years for lung cancer outcomes. Individuals not qualifying by either criteria will not be offered screening, but samples of them will be followed for lung cancer outcomes. Outcomes in discordant groups, USPSTF+ve/PLCOm2012-ve and USPSTF-ve/PLCOm2012+ve, are informative. Numbers of lung cancers, abnormal suspicious for lung cancer scans (a marker of future lung cancers) and individuals enrolled, and sensitivity and specificity and positive predictive values of the two criteria will be compared.
4c3880bb027f159e801041b1021e88e8 Result
Results
As of March 2018, ILST centers in Canada (British Columbia and Alberta), Australia, and the United Kingdom had enrolled and scanned 1938 individuals. Study results are summarized in Figure 1.
8eea62084ca7e541d918e823422bd82e Conclusion
Conclusion
Interim analysis of ILST data, suggests that classification accuracy of lung cancer screening outcomes support the PLCOm2012 criteria over the USPSTF criteria. Individuals who are USPSTF+ve and PLCOm2012-ve appear to be at such low baseline risk (0.46%) that they may be unlikely to benefit from screening.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
P2.11 - Screening and Early Detection (Not CME Accredited Session) (ID 960)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/25/2018, 16:45 - 18:00, Exhibit Hall
-
+
P2.11-29 - Impact of an Information-Film to Promote Informed Decision-Making in Individuals Taking Part in a Lung Cancer Screening Demonstration Pilot (ID 12757)
16:45 - 18:00 | Author(s): Angshu Bhowmik
- Abstract
Background
Lung cancer screening by Low Dose CT (LDCT) is underway in the United States, where a shared decision-making process is mandated for insurer funding. The potential harms of screening are complex and difficult to communicate. Participants do not always read written materials and audio-visual aids have been shown to improve informed decision-making in other areas of medicine. There are limited studies on the use of decision aids in lung cancer screening.
a9ded1e5ce5d75814730bb4caaf49419 Method
A five-minute information-film was made to explain the benefits and risks of lung cancer screening. Qualitative research informed the content and format to make it accessible for individuals of varying demographic and educational backgrounds. A sub-sample of participants (n=229) from a lung cancer screening pilot were randomised to watch the film and read a written information booklet (Group A) or read the booklet alone (Group B). Objective and subjective knowledge of the risks and benefits of screening were assessed before and after the intervention, and again after discussion with a heath care professional, when decisional conflict and satisfaction were also assessed.
4c3880bb027f159e801041b1021e88e8 Result
120 and 109 participants were randomised to groups A and B respectively. There was a statistically significant improvement in subjective and objective knowledge scores post-administration of the intervention in both groups (p<0.001). In a multivariate analysis adjusted for educational level and baseline knowledge score, post interventional scores were independently associated with film group (p=0.036). There were no group differences in decisional conflict or satisfaction, or in subsequent attendance for LDCT. Participants’ views on the acceptability of the film are presented in table 1.
Table 1: Feedback and acceptability of information film
8eea62084ca7e541d918e823422bd82e Conclusion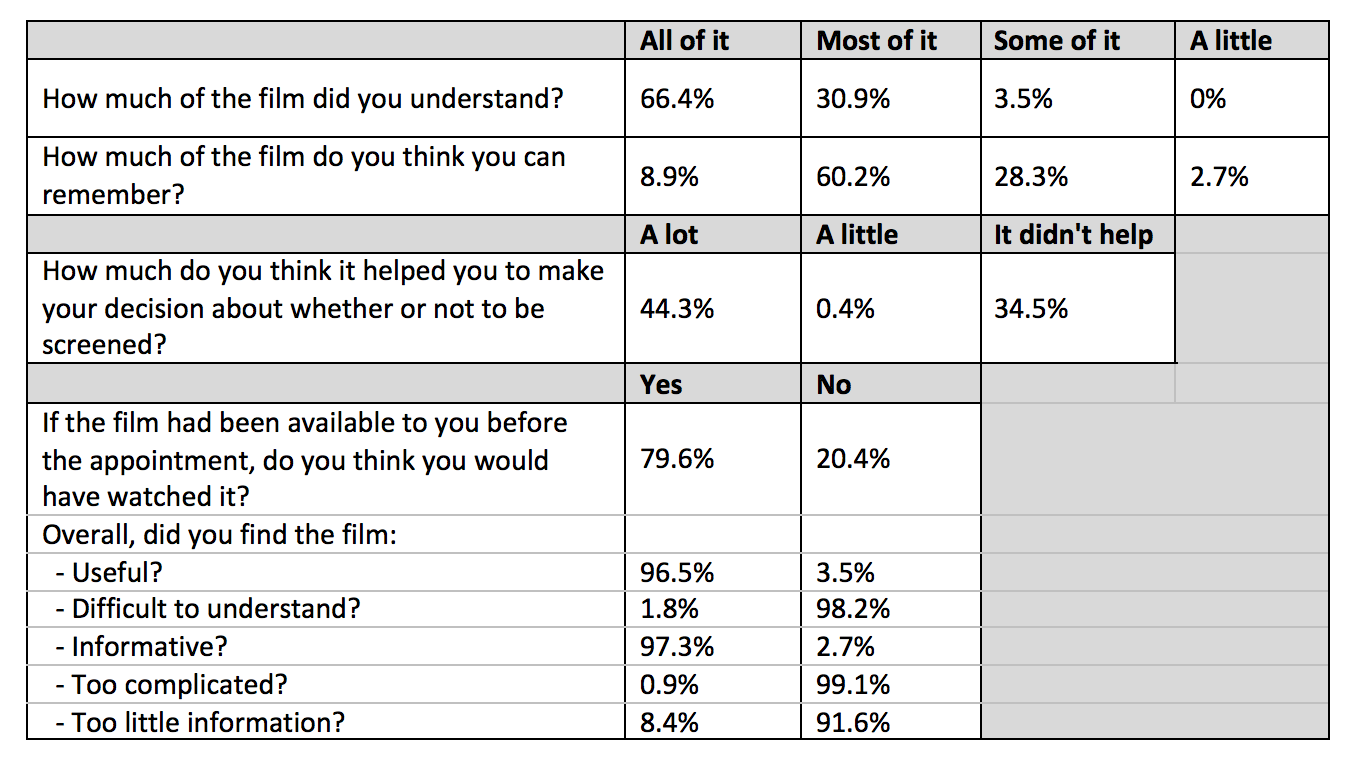
The information-film was well-accepted and increased knowledge scores more than a written booklet alone, without raising decisional conflict or reducing attendance for LDCT.
6f8b794f3246b0c1e1780bb4d4d5dc53


