Virtual Library
Start Your Search
Lizza Hendriks
Author of
-
+
MA08 - Clinical Trials in Brain Metastases (ID 906)
- Event: WCLC 2018
- Type: Mini Oral Abstract Session
- Track: Advanced NSCLC
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 15:15 - 16:45, Room 203 BD
-
+
MA08.09 - Impact of Brain Metastases in Immune Checkpoint Inhibitors (ICI) Treated Advanced Non-Small Cell Lung Cancer (NSCLC) Patients (ID 12575)
16:10 - 16:15 | Presenting Author(s): Lizza Hendriks
- Abstract
- Presentation
Background
Brain metastases (BM) are frequent in NSCLC. Unfortunately, patients with (untreated) BM are often excluded from ICI trials so that their outcome on ICI is largely unknown..
a9ded1e5ce5d75814730bb4caaf49419 Method
Retrospective data collection of all consecutive advanced ICI treated NSCLC patients in 6 centers (5 French, 1 Dutch) (nov 2012 – march 2018). Active BM was defined as non-irradiated new and/or growing lesions on brain imaging < 6 weeks before ICI start. Progression free survival (PFS), overall survival (OS) and site of progression on ICI was collected.
4c3880bb027f159e801041b1021e88e8 Result
945 patients included: 63% male, 83% WHO PS 0-1, median age 64 years, 73% non-squamous, 4% targetable driver mutations, 33% known PD-L1 (65% ≥1% expression). ICI treatment was median 2nd line (range 1-12), 94% had monotherapy PD-(L)1 inhibition. 241 patients (26%) had BM, 68% had previous cranial irradiation, 40% had active BM. BM patients were significantly younger than others (61 vs 66 years), had more adenocarcinoma (78 vs 62%), more organs involved (median 3 vs 2), a poorer PS (0-1: 76 vs 85%) and more steroids at baseline (26 vs 9%). Median follow-up: 15 months. Median (95% CI) PFS and OS without and with BM were 2 (2-3) vs 2 (1-2) months and 13 (9-16) vs 9 (7-13) months, respectively. In multivariate analysis, > 2 metastatic sites, PS ≥2 and steroids use were associated with worse PFS and OS, BM were not (table 1). In univariate analysis of BM patients, active BM were not associated with worse outcome compared to stable BM (HR PFS 0.98 (p=0.66), HR OS 0.93 (p=0.92)). Progressing BM patients had more often brain PD and a dissociated response (not specifically brain dissociated) on ICI (40 vs 12% and 13 vs 7%, respectively).
Factor PFS HR (95% CI) p-value OS HR (95% CI) p-value Age > 65 vs ≤ 65 1.02 (0.87-1.20) 0.79 1.11 (0.92-1.34) 0.29 Smoking yes vs no 0.53 (0.41-0.69) <0.001 0.81 (0.59-1.12) 0.20 Histology squamous vs adeno 1.07 (0.89-1.28) 0.78 1.24 (0.99-1.55) 0.12 Nr of organs with metastases > 2 vs ≤ 2 1.28 (1.09-1.50) 0.003 1.48 (1.22-1.80) <0.001 Immuno line > 2 vs ≤ 2 1.11 (0.94-1.30) 0.22 1.10 (0.91-1.33) 0.34 WHO PS 0-1 vs ≥2 2.14 (1.75-2.62) <0.001 3.48 (2.78-4.36) <0.001 Use of corticosteroids yes vs no 1.36 (1.10-1.69) 0.005 1.31 (1.03-1.68) 0.03 BM yes vs no 1.05 (0.88-1.26) 0.58 0.96 (0.77-1.19) 0.70
8eea62084ca7e541d918e823422bd82e Conclusion
BM, treated or active, do not negatively impact outcome on ICI although BM failure is more common in these patients.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
MA25 - Oligometastasis: Defining, Treating, and Evaluating (ID 929)
- Event: WCLC 2018
- Type: Mini Oral Abstract Session
- Track: Oligometastatic NSCLC
- Presentations: 3
- Moderators:
- Coordinates: 9/26/2018, 13:30 - 15:00, Room 203 BD
-
+
MA25.01 - EORTC Lung Cancer Group Survey to Define Synchronous Oligometastatic Disease in NSCLC (ID 13770)
13:30 - 13:35 | Presenting Author(s): Lizza Hendriks
- Abstract
- Presentation
Background
Synchronous oligometastasic disease (sOMD) has been described as a separate disease entity; however there is no consensus on what specific criteria constitutes sOMD in NSCLC. A consensus group (CG) was formed aiming to agree on a common sOMD definition (sOMD-d) that could be used in future clinical trials. A European survey was circulated to inform the discussion on sOMD-d.
a9ded1e5ce5d75814730bb4caaf49419 Method
An EORTC Lung Cancer Group (LCG) / sOMD-d CG survey containing 31 questions on sOMD-d was distributed between 14/12/17 and 19/02/18 to EORTC LCG, sOMD-d CG, and several European thoracic oncology societies’ members.
4c3880bb027f159e801041b1021e88e8 Result
444 responses were analyzed (radiation oncologist: 55% [n=242], pulmonologist: 15% [n=66], medical oncologist: 14% [n=64]; 78% with >5 years’ experience in treating NSCLC). Belgium (14%, n=62), Italy (12%, n=55), Germany (11%, n=47), and Netherlands (10%, n=44) contributed most. 81% (n=361) physicians aimed to cure sOMD NSCLC patients and 82% (n=361) included the possibility to treat the patient with radical intent in their sOMD-d. The maximum number of metastases considered in sOMD-d varied: 19%, 42%, 4%, and 17% replied <2, 3, 4, and >5 metastases, respectively. 79% (n=353) stated that the number of organs involved was important for sOMD-d, and most (80%, n=355) considered that only <3 involved organs (excluding primary) should be included in the definition. 317 (71.7%) allowed mediastinal lymph node involvement (MLN) in the sOMD-d, and 22.1% of them counted MLN as a metastatic site. For 195/327 (60%), when N2/N3 disease is included in the sOMD-d, there is no specific issue regarding the MLN volume/location as long as radical treatment is possible. 384 (86%) considered pulmonary metastasis (outside primary tumor: M1a) as metastatic site. Most physicians confirmed sOMD patients with brain MRI (91%, n=403) and PET-CT (98%, n=437). For mediastinum staging, most (64%, n=285) respondents stated that histology/cytology should be obtained when PET-CT shows suspected lymph nodes or in case of a central primary tumor. Pathology proof of metastatic disease was necessary in sOMD for 315 (71%) physicians, and 37% (n=163) acknowledged that histology should be obtained from at least from one metastatic site. Preferred primary outcome parameter in clinical trials of sOMD was overall survival (73%, n=325).
8eea62084ca7e541d918e823422bd82e Conclusion
Although certain consensual answers were obtained (81% aimed to cure and >90% mandated baseline imaging with PET-CT and brain MRI), a number of issues remain unresolved and will require further discussion by a panel of experts to agree on a sOMD-d.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
MA25.02 - Searching for a Definition of Synchronous Oligometastatic (sOMD)-NSCLC: A Consensus from Thoracic Oncology Experts (ID 13452)
13:40 - 13:45 | Author(s): Lizza Hendriks
- Abstract
- Presentation
Background
Recent prospective single centre studies reported improved outcomes in patients with sOMD-NSCLC who were treated with radical intent. Since then sOMD has been perceived as a separate disease entity. However, a clear definition of sOMD-NSCLC is lacking. We aimed to develop a definition and diagnostic criteria of sOMD-NSCLC following a consensus process.
a9ded1e5ce5d75814730bb4caaf49419 Method
A European multidisciplinary consensus group was established with representatives from different scientific societies. Consensus questions were extracted from a survey, case series and a systematic review. The questions were discussed, and the statement formulated during a consensus meeting in Dublin (23.01.18).
4c3880bb027f159e801041b1021e88e8 Result
Summary of consensus statement
Defining sOMD-NSCLC
Definition of sOMD is relevant for patients in whom a radical treatment is technically feasible with acceptable toxicity, taking into account all sites, that may modify the course of the disease leading to a long-term disease control.
All sites must be technically and safely treatable.
The maximum number of metastases/organs meeting the criteria involved will depend on the possibility of offering a treatment strategy with radical intent, taking into account local control and toxicity. Based on the systematic review, a maximum of 5 metastases and 3 organs is proposed.
Diffuse serosal metastases and bone marrow involvement are excluded.
Mediastinal lymph node (MLN) involvement should be considered as locoregional disease in the definition of sOMD-NSCLC.
MLN involvement is of importance in determining if a radical local treatment of the primary tumour may be applied and the MLN will not be counted as a metastatic site.
Staging of sOMD-NSCLC
PET-CT and brain imaging are considered mandatory.
In case of a solitary liver metastasis a dedicated MRI of the liver and for a solitary pleural metastasis, thoracoscopy and biopsies of distant ipsilateral pleural sites are advised.
Staging of the mediastinum requires a minimum of a FDG-PET scan, with pathological confirmation preferred if this influences the treatment strategy.
Pathological proof is required unless the MDT decides that the risk outweighs the benefit. Pathology proof is advised for single metastatic location and if it may change the therapeutic strategy, confirmation of the MLN involvement is recommended.
8eea62084ca7e541d918e823422bd82e Conclusion
A multidisciplinary consensus statement on the definition and staging of sOMD-NSCLC was formulated taking into account results of a European survey, a systematic review and case discussion. This statement might be helpful to standardise inclusion criteria in future clinical trials. However, the definition of sOMD may change over time when more prospective data will become available.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
MA25.03 - Defining Oligometastatic Non-Small Cell Lung Cancer (NSCLC): An Evolving Multidisciplinary Expert Opinion (ID 12573)
13:35 - 13:40 | Presenting Author(s): Lizza Hendriks
- Abstract
- Presentation
Background
Synchronous oligometastatic NSCLC definition varies between: 1 metastasis in 1 organ (TNM8), 1-3 metastases (ESMO), ≤3 metastases after systemic treatment with mediastinal nodes (MLN) counting as 1 site (Gomez, Lancet Oncol 2016) to 3-≥5 metastases in ongoing trials. A single definition is however needed to design and compare trials. To assess synchronous oligometastatic NSCLC definitions used by clinical experts in daily practice and its evolution, we redistributed a 2012-case based survey (Dooms et al, presented at WCLC 2013).
a9ded1e5ce5d75814730bb4caaf49419 Method
In December 2017, 10 real-life multidisciplinary team (MDT) discussed patients (all good condition, no significant comorbidities, 18FFDG-PET and brain MRI staged, all < 5 metastases, 9/10 ≤ 3 metastases, oncogene-addicted or wildtype NSCLC) were distributed to 33 international NSCLC experts involved in the EORTC oligometastatic NSCLC consensus group, questioning: 1) can you discuss these cases in your MDT?, 2) do these patients have oligometastatic disease? and 3) what is your treatment proposal for the oligometastatic disease patients? Current answers were compared to the previous ones, and the real-life treatment and survival of the patients was added.
4c3880bb027f159e801041b1021e88e8 Result
26/33 experts (24 centers) replied: 8 medical oncologists, 7 pulmonologists, 7 radiation oncologists, 4 thoracic surgeons. 62% discussed the cases in their MDT. 1 case had 100% oligometastatic disease consensus, 3 cases had > 90% consensus, the number of treatment proposals varied between 3 to 8 (Table). Radical treatment was more often offered in case of a single metastasis or N0 status. Compared to 2012 there was a trend towards a more conservative oligometastatic definition and chemotherapy was more often included in the treatment proposal.
table 1 Case TNM8 oligometastatic
yes answer %
2012 / 2017Number of tx
proposals
2012 / 2017
Radical tx
answers %
2012/2017Real life radical
tx intent
real life survival
(months) /
5Y survival
EGFR+ T2aN3M1c (3 brain mets) 55 / 38 2 / 5 27 / 23 - 40.1 / - EGFR+ T4N0M1a (ground glass) 36 / 35 4 / 3 45 / 35 + 65.2 / + T2aN1M1b (solitary renal) 91 / 96 5 / 5 100 / 92 + 8.3 / - T1bN3M1b (solitary adrenal) 73 / 58 4 / 5 36 / 54 + 66.1 / + T2bN1M1c (adrenal + pelvic node) 55 / 50 2 / 5 36 / 46 - 18.6 / - T2aN0M1c (3 liver mets) 64 / 69 4/ 5 27 / 62 - 51.5 / - T2aN2M1b (solitary bone) 91 / 92 4 / 5 73 / 85 + 13.4 / - T3N1M1c (2 brain mets) 91 / 96 3 / 8 73 / 85 + 39.6 / - T2aN0M1c (1 lung, 1 pancreas) 82 / 69 5 / 4 64 / 50 + 74.0 / + T1bN0M1b (solitary bone) 100 / 100 3 / 5 82 / 92 + 11.6 / -
8eea62084ca7e541d918e823422bd82e Conclusion
Synchronous oligometastatic NSCLC definition was more conservative than in 2012 and linked to radical intent of treatment. Number of organs, MLN status and possibility for radical treatment seem to be components of daily practice synchronous oligometastatic definition.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
OA07 - Oligometastasis: What Should Be the State-Of-The-Art? (ID 905)
- Event: WCLC 2018
- Type: Oral Abstract Session
- Track: Oligometastatic NSCLC
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 15:15 - 16:45, Room 107
-
+
OA07.07 - PFS and OS Beyond 5 years of NSCLC Patients with Synchronous Oligometastases Treated in a Prospective Phase II Trial (NCT 01282450) (ID 13389)
16:20 - 16:30 | Author(s): Lizza Hendriks
- Abstract
- Presentation
Background
There is increasing interest in the treatment of synchronous oligometastases of NSCLC. Two randomized studies demonstrated an increased PFS by adding a radical local treatment to systemic therapy in responding patients, but long-term data are lacking. We previously reported a median PFS of 12 months and a median OS of 13.5 months in 39 radically treated patients with synchronous oligometastases in a prospective study (De Ruysscher J Thorac Oncol 2012). As the minimal follow-up is now exceeding 6 years, we here report the long-term PFS and OS.
a9ded1e5ce5d75814730bb4caaf49419 Method
Prospective single-arm phase II trial. The main inclusion criteria were pathologically proven NSCLC stage IV with less than five metastases at primary diagnosis, amendable for radical local treatment (surgery or radiotherapy). No previous response to systemic treatment was required.
4c3880bb027f159e801041b1021e88e8 Result
Forty patients were enrolled, 39 of whom were evaluable (18 men, 21 women); mean age was 62.1 ± 9.2 years (range, 44-81). Twenty-nine (74%) had local stage III; 17 (44%) brain, seven (18%) bone, and four (10%) adrenal gland metastases. Thirty-five (87%) had a single metastatic lesion. Thirty-seven (95%) of the patients received chemotherapy as part of their primary treatment.
Median overall survival (OS) was 13.5 months (95% CI 7.6-19.4); 1-, 2-, 3-, 4-, 5, 6-year OS was 56.4%, 23.3%, 12.8 %, 10.3 %, 7.7 %, 5.1 % (2 patients), respectively.
Median progression-free survival (PFS) was 12.1 months (95% CI 9.6-14.3); 1-, 2-, 3-, 4-, 5, 6-year PFS was 51.3%, 13.6 %,12.8 %, 7.7 %, 7.7 %, 2,5 % (1 patient), respectively.
Of the 3 patients with a PFS after 5 years, 1 had a squamous cell cancer T2N2 with a single pathologically proven bone metastasis in the sternum, 1 had a NSCLC-NOS T4N0 with a single adrenal metastasis, and 1 a T1N2 adenocarcinoma with a pathologically proven contralateral lung metastasis. The latter patient is still free of disease.
Two patients developed a second primary cancer: 1 tongue carcinoma after 70 months and 1 an adenocarcinoma in the contralateral lung after 71 months. Both patients died of their second cancer.
Three patients (7.7 %) had a local recurrence, all in the PTV of their primary tumor.
Only one patient was treated with a TKI (gefitinib) at progression.
8eea62084ca7e541d918e823422bd82e Conclusion
After radical treatment of oligometastases, approximately 8 % of the patients achieve a PFS after 5 years. Entering patients in trials combining local therapy with novel systemic agents (e.g. chemo-immunotherapy) remains mandatory.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
P1.01 - Advanced NSCLC (Not CME Accredited Session) (ID 933)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 3
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.01-07 - Immune-Related Pneumonitis in NSCLC Patients Treated with Immune Checkpoint Inhibitors (ICI): Impact of Previous Thoracic Radiotherapy (ID 12805)
16:45 - 18:00 | Author(s): Lizza Hendriks
- Abstract
Background
Pneumonitis is a potentially lethal side effect of immune checkpoint inhibitors (ICI), occurring in 1–5% of patients enrolled in clinical trials. Little is known about the interactions between ICI and previous thoracic radiation. This is the aim of the present study.
a9ded1e5ce5d75814730bb4caaf49419 Method
Between December 2012 and November 2017, 318 consecutive non-small cell lung cancer (NSCLC) patients received ICI in our Institution and their charts were retrospectively analyzed. Primary endpoint was to determine whether previous radiotherapy had an effect on pulmonary toxicity. Pulmonary toxicity was retrospectively assessed by Common Terminology Criteria for Adverse Events version 4.0.
4c3880bb027f159e801041b1021e88e8 Result
Median follow-up was 32.8 months [95%CI: 5-190]. Median age at the start of ICI was 63 years. 205 patients (64,5%) were males, 103 (32,4%) smokers and 250 (78,6%) with PS ≤1; 206 (64,8%) had adenocarcinoma and 76 (23,9%) squamous; 79 (24,8%) were KRAS mutated, 18 (5,5%) EGFR mutated and 5 (1,6%) ALK positive. PDL1 was ≥ 1% by immunohistochemistry in 86 (27%), negative in 37 (11,6%) and unknown in 196 (61,3%) patients. ICI treatment was median 3rd line (range: 1-12), 89,4% monotherapy PD-(L)1 inhibition.
72 patients (22,6%) received a thoracic RT: 62 out of the 72 RT patients (87,5%) were irradiated with a curative intent. 53 patients (73,6% of the RT patients) received thoracic 3D-conformal RT or intensity modulated RT (normo- or mildly hypofractionated), whereas 9 received SBRT.
16,7% of the RT patients (12/72) showed a G1-4 immune-related pneumonitis (with a G=>3 of 11,1%), whereas for never-irradiated patients the G1-3 rate of immune-related pneumonitis was 2,4% (6/246), with only 1 G3 toxicity observed and no G>4 (t-test, p 0,001).
Median interval between the onset of the immune-related pneumonitis and the end of the RT was 22,4 months.
8eea62084ca7e541d918e823422bd82e Conclusion
NSCLC patients treated with ICI may be at higher risk of developing immune-related pneumonitis when previously treated with curative-intent thoracic RT.
6f8b794f3246b0c1e1780bb4d4d5dc53 -
+
P1.01-18 - Immunosenescence Correlates with Progression upon PD-(L)-1 Blockade (IO) in Advanced Non-Small Cell Lung Cancer (aNSCLC) Patients. (ID 14074)
16:45 - 18:00 | Author(s): Lizza Hendriks
- Abstract
Background
Immunosenescence is a progressive remodeling of immune functions with a multifactorial etiology (i.e. aging, chronic inflammation, cancer). Although a CD28-CD57+KLRG1+ phenotype on peripheral T-lymphocytes is a potential hallmark of immunosenescence, the characterization of such phenotype in IO-treated NSCLC patients and the correlation with clinical characteristics and benefit from immunotherapy are unknown.
a9ded1e5ce5d75814730bb4caaf49419 Method
A senescent immune phenotype (SIP) defined as a percentage of circulating CD8+CD28-CD57+KLRG1+ T-lymphocytes was assessed by flow cytometry (FC) on fresh blood samples from IO-treated aNSCLC patients (03/2017–04/2018). A log-rank maximization method was used to identify a SIP cut-off level and dichotomize patients accordingly. The objective was to correlate SIP with clinical characteristics and RECIST response by univariate logistic regression analysis.
4c3880bb027f159e801041b1021e88e8 Result
39 aNSCLC patients were evaluable for SIP before IO: 38% ≥ 65 years, 87% non-squamous, 38% KRAS mutated, 54% with PD-L1 expression ≥1%, 13% chemotherapy naïve. Among 30 patients evaluable for IO response, 53% had progression (PD), 27% stability (SD), 20% partial response (PR). Median PFS was 1.9 months (95% CI 1.5; 2.5). OS was not calculated due to the short follow-up [6 months (95% CI 4-11)]. SIP (% CD28-CD57+KLRG1+) median value on circulating CD8+ lymphocytes was 15.26% (min 1.87%, max 56.28%). Overall, 13 (33%) of 39 patients had >22.25% CD8+ lymphocytes with a CD28-CD57+KLRG1+ phenotype, being classified as SIP+. SIP status did not correlate with age, IO-baseline patients’ characteristics or chemotherapy exposure. Among patients evaluable for IO response, only 1 (10%) of 10 SIP+ experienced disease control (PR/SD), compared to 13 (65%) of 20 SIP- patients; similarly, PD rate was significantly higher in SIP+ compared to SIP- patients (90% vs 35%, p=0.007) (Figure).
8eea62084ca7e541d918e823422bd82e Conclusion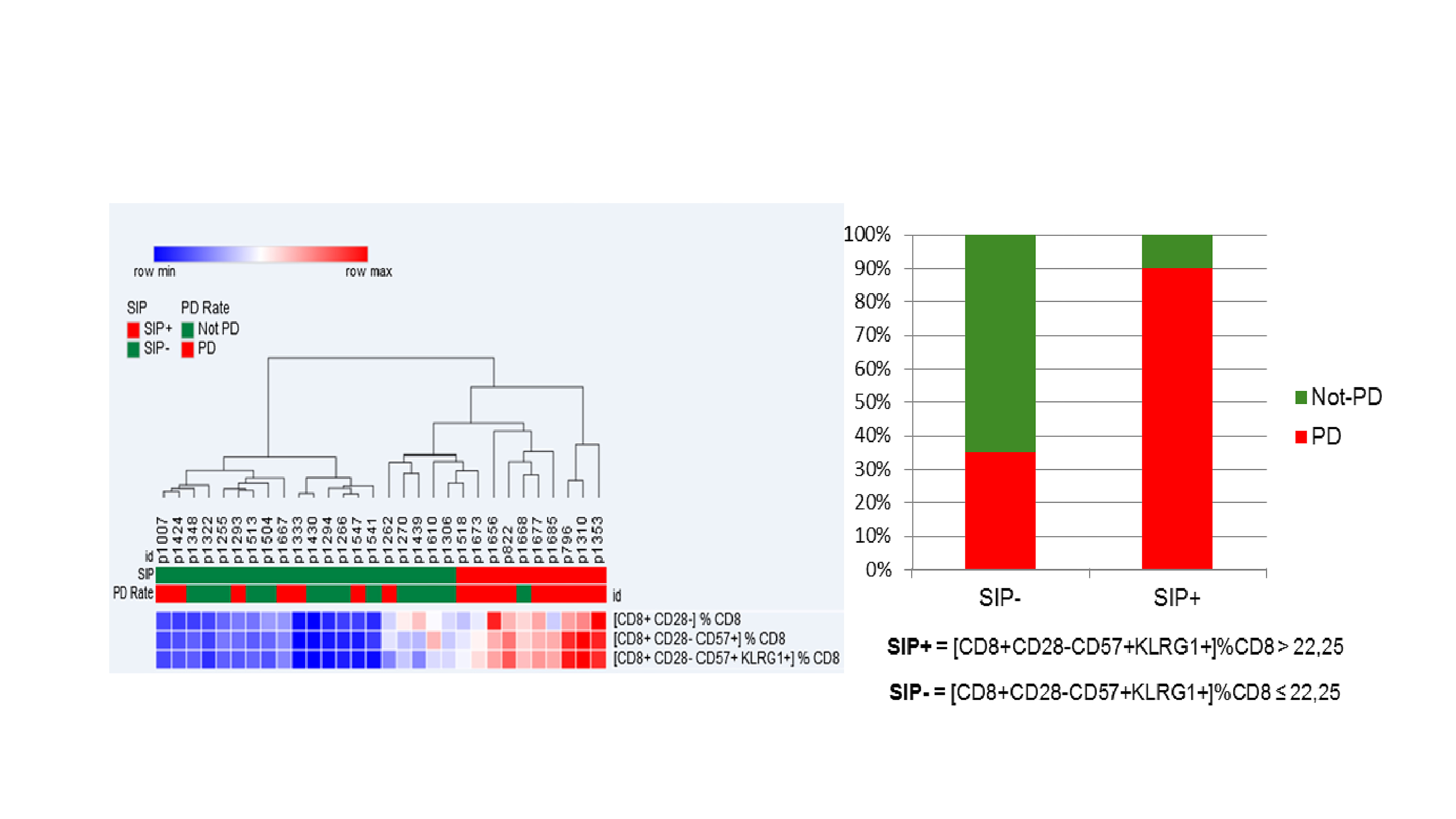
Immunosenescence, monitored by FC measurement of 3 surface molecules on circulating CD8 + lymphocytes, is observed in 33% of aNSCLC patients, is independent of age and correlates with lower IO disease control rate.
6f8b794f3246b0c1e1780bb4d4d5dc53 -
+
P1.01-68 - Correlation of the Lung Immune Prognostic Index (LIPI) and PDL1 Status with Outcomes for Immune Checkpoint Inhibitors in Advanced NSCLC Patients (ID 14256)
16:45 - 18:00 | Author(s): Lizza Hendriks
- Abstract
Background
Baseline LIPI, based on derived NLR (neutrophils/[leucocytes-neutrophils]) and lactate dehydrogenase (LDH) was associated with outcomes for immune checkpoint inhibitors in advanced NSCLC patients. We assessed the correlation between LIPI and PDL1 for ICI outcomes in NSCLC.
a9ded1e5ce5d75814730bb4caaf49419 Method
Baseline dNLR and LDH and clinical data were retrospectively collected in advanced NSCLC patients, treated with PD1/PDL1 +/- CTLA4 inhibitors from Nov. 2012 to Mar. 2018, in a multicentric cohort (N=794) from 11 centers. LIPI stratified 3 groups: good (dNLR<3+LDH<upper limit of normal (ULN), intermediate (dNLR>3 or LDH>ULN), poor risk (dNLR>3+LDH>ULN). PDL1 positivity was defined as ≥ 1% tumor cells expression by immunohistochemistry.
4c3880bb027f159e801041b1021e88e8 Result
476 patients (60%) were male, 693 (87%) smokers, 695 (88%) had PS ≤1, with median age 65; 576 (73%) had nonsquamous histology. PDL1 was ≥ 1% in 195 (70%) patients, negative in 82 (30%), and unknown in 517. The median of prior lines was 1 (0-11). The median PFS and OS were 4 months (m) [95% CI 4-5] and 12 m [10-15]. dNLR was>3 in 276 (35%) and LDH>ULN in 290 (37%) patients. LIPI stratified 349 patients as good (44%), 323 (41%) as intermediate and 121 (15%) as poor LIPI risk groups. LIPI was an independent factor for OS (table) and PFS (HR 2.58; CI 1.3-5.2, P=0.02). ≥ 1% PDL1 and ≥ 50% PDL1 were not correlated with OS and PFS. Median OS for good, intermediate, and poor LIPI risk groups were 21 m [17-23], 11 m [9-14] and 4 m [2-6], respectively (P=<0.0001). Median PFS for good, intermediate, and poor risk was 5 m [5-7], 4 m [3-5], and 2 m [1-3], respectively (P=0.0005). No differences were observed in LIPI groups according to the PDL1 expression.
8eea62084ca7e541d918e823422bd82e ConclusionMultivariate analysis for OS
HR
95% CI
P value
Immunotherapy line
>2
2.117
0.641
6.992
0.219
N# Metastasis sites
≥2
1.242
0.727
2.121
0.428
Performance status
≥2
2.141
1.059
4.332
0.034
Albumin
>35 g/dL
0.867
0.507
1.48
0.6
LIPI
Intermediate
Poor
1.697
4.178
0.917
1.956
3.142
8.925
0.001
PDL1 IHC
≥1%
0.713
0.406
1.252
0.239
Baseline LIPI is associated with ICI outcomes in advanced NSCLC, regardless the PDL1 expression. LIPI should be evaluated in prospective clinical trials.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P3.12 - Small Cell Lung Cancer/NET (Not CME Accredited Session) (ID 978)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/26/2018, 12:00 - 13:30, Exhibit Hall
-
+
P3.12-11 - Association of the Lung Immune Prognostic Index (LIPI) with Outcomes for Immune Checkpoint Inhibitors in Diffuse SCLC Patients (ID 14200)
12:00 - 13:30 | Author(s): Lizza Hendriks
- Abstract
Background
Pretreatment LIPI (Lung Immune Prognostic Index), based on derived NLR (neutrophils/[leucocytes-neutrophils] ratio) and lactate dehydrogenase (LDH) has been associated with outcomes for immune checkpoint inhibitors (ICI) in advanced NSCLC patients. We tested whether LIPI has the same role in diffuse small cell lung cancer (SCLC) patients.
a9ded1e5ce5d75814730bb4caaf49419 Method
Baseline dNLR and LDH and clinical data were retrospectively collected in SCLC patients, treated with ICI (PD1 inhibitor, PDL1 inhibitors +/- CTLA4 inhibitor) from April 2014 to Jan. 2018 (N=66) from 6 European centers. LIPI was calculated combining dNLR and LDH, stratifying 3 risk groups: good (dNLR<3+LDH<upper limit of normal (ULN), intermediate (dNLR>3 or LDH>ULN), poor (dNLR>3+LDH>ULN). The primary endpoint was overall survival (OS), and secondary endpoint was progression-free survival (PFS).
4c3880bb027f159e801041b1021e88e8 Result
Fifty-three patients (80%) were males, 58 (88%) smokers and all patients had PS ≤1, with median age 63 years (41-82). PDL1 was ≥ 1% by immunohistochemistry in 6 patients, and unknown in 60 patients. The median of prior lines was 1 (0-6). Platinum-based therapy was the prior line in 63 (95%) patients, with ORR of 88%. The median PFS and OS with ICI were 2.7 months (m) [95% CI 1.87-4.43] and 10.3 m [95% CI 5.8-12.6]. dNLR was greater than 3 in 16 (25%) and LDH> Upper Limit of Normal (ULN) in 33 (50%) patients. Based on both, LIPI stratified the population in 3 groups: 26 patients as good (40%), 29 (45%) as intermediate and 10 (15%) as poor LIPI risk groups. LIPI was an independent factor for OS (HR 2.77, 95% CI 1.07-7.14, P=0.03) and PFS (HR 3.13, 1.37-7.16, P=0.01). Median OS for good, intermediate, and poor risk groups were 11.4 m [95% CI 5.5-27.3], 11 m [95% CI 6.8-not-reached (NR)] and 2.3 m [95% CI 0.7-NR], respectively (P=0.004). Median PFS for good, intermediate, and poor risk groups were 3 m [95% CI 1.9-12.6], 2.8 m [95% CI 1.6-6.0 and 1.2 m [95% CI 0.47-NR], respectively (P=0.004).
8eea62084ca7e541d918e823422bd82e Conclusion
Baseline LIPI poor risk group is associated with poor outcomes for ICI in diffuse SCLC patients. LIPI effect in a validation cohort is currently evaluated.
6f8b794f3246b0c1e1780bb4d4d5dc53


