Virtual Library
Start Your Search
Alexander Drilon
Author of
-
+
MA04 - Novel Approaches with IO (ID 900)
- Event: WCLC 2018
- Type: Mini Oral Abstract Session
- Track: Immunooncology
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 13:30 - 15:00, Room 107
-
+
MA04.03 - Immunotherapy for Non-Small Cell Lung Cancers (NSCLC) with Oncogenic Driver Mutations: New Results from the Global IMMUNOTARGET Registry (ID 13187)
13:40 - 13:45 | Author(s): Alexander Drilon
- Abstract
- Presentation
Background
Prospective data on immunotherapy for NSCLC with oncogenic driver mutations are limited. We recently reported first results from the global IMMUNOTARGET registry (Mazières, ASCO 2018). Here, we present new data for PD-L1 and mutation subgroups.
a9ded1e5ce5d75814730bb4caaf49419 Method
In 2017, we started an international retrospective registry study ("IMMUNOTARGET") for patients with advanced NSCLC, known driver mutations (KRAS, EGFR, ALK, ROS1, BRAF, HER2, MET and RET) and PD-L1 immune checkpoint inhibitor therapy. The registry is approved by University of Toulouse and Swissethics, and funded by University of Toulouse and Cantonal Hospital of Lucerne. Anonymized real-world data submitted to the coordinating center include: patient and tumor characteristics, mutation test methods and results, systemic therapy lines, immune related adverse events, best response by RECIST, survival, and tumor PD-L1 expression (optional). Statistical calculations including best response, median PFS and OS are done at University of Toulouse.
4c3880bb027f159e801041b1021e88e8 Result
In April 2018, the registry included 551 pts from Europe, USA, Israel and Australia. Patients were 50% male/female, 28% current smokers, median age 60 years (range 28-83), 85% had PS0/1. Most (73%) tumors were stage IV at diagnosis, almost all (96%) were adenocarcinomas. Molecular classification by dominant driver mutation: KRAS=271 (49%), EGFR=125 (23%), BRAF=43 (8%), MET=36 (7%), HER2=29 (5%), ALK=23 (4%), RET=16 (3%), ROS1=7 (1%), 1 (0.2%) not classified (ALK+RET+MET). Most pts received nivolumab (466) or pembrolizumab (48) and were treated with immunotherapy in second or third line (67%). The median number of cycles was 5 (range 1-68). Fifty (11%) pts had grade 3-5 toxicity. Median OS from start of immunotherapy was 13.3 months, median PFS was 2.8 months. Best response was PR/CR in: KRAS=26%, BRAF=24%, ROS1=17%, MET=16%, EGFR=12%, HER2=7%, RET=6%, ALK=0%. Percentage of PD-L1 positive cells was available for 177 pts: 0%=71 (40%), 1-49%=46 (26%), 50-100%=60 (34%). Median % of positive cells was highest for ROS1 (90%), BRAF (50%), MET (30%) and RET (26%) mutant tumors. PD-L1 positivity was predictive for improved PFS in KRAS and EGFR mutant tumors. PD-L1 status was known in 18 tumors with ALK, ROS1 or RET rearrangements: 5 had 0%, 4 had 1-49% and 9 had 50%-100%. No tumor remissions were observed in this subgroup. The registry remains open, updated results will be presented at the conference.
8eea62084ca7e541d918e823422bd82e Conclusion
Although response rates were lower than in KRAS mutant NSCLC, individual tumors with other driver mutations responded to immunotherapy. PD-L1 expression may not accurately predict clinical benefit from immunotherapy in some molecular subgroups, better markers are needed.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
OA02 - Novel Therapies in ROS1, HER2 and EGFR (ID 893)
- Event: WCLC 2018
- Type: Oral Abstract Session
- Track: Targeted Therapy
- Presentations: 2
- Moderators:
- Coordinates: 9/24/2018, 10:30 - 12:00, Room 105
-
+
OA02.01 - Efficacy and Safety of Entrectinib in Locally Advanced or Metastatic ROS1 Fusion-Positive Non-Small Cell Lung Cancer (NSCLC) (ID 13903)
10:30 - 10:40 | Author(s): Alexander Drilon
- Abstract
- Presentation
Background
Entrectinib is a central nervous system (CNS) active, potent, and selective inhibitor of ROS1, TRKA/B/C and ALK. Entrectinib is more potent against ROS1 than crizotinib, the only agent currently approved for the treatment of ROS1-positive NSCLC. Interim data demonstrated that entrectinib was tolerable and achieved high objective response rates (ORR) in patients with ROS1-positive, ROS1 inhibitor-naive NSCLC, including patients with baseline CNS disease (Ahn MJ WCLC 2017).
a9ded1e5ce5d75814730bb4caaf49419 Method
Phase 1/2 studies of entrectinib (ALKA, STARTRK-1, STARTRK-2; EudraCT 2012-000148-88; NCT02097810; NCT02568267) enrolled patients with locally advanced or metastatic solid tumors. The safety-evaluable population included patients who received ≥1 dose of entrectinib. The integrated efficacy analysis included ROS1-positive NSCLC patients enrolled based on identification of ROS1 fusions via nucleic acid-based diagnostic platforms. Safety was assessed by monitoring adverse events (AEs), laboratory tests, and physical examination. Tumor assessments were performed at the end of cycle 1 and every 8 weeks thereafter. All scans were submitted for blinded independent central review (BICR) using RECISTv1.1. Primary endpoints were ORR and duration of response (DOR) by BICR. Key secondary objectives were progression-free survival (PFS), overall survival (OS), and safety. Additional endpoints evaluated in patients with baseline CNS disease were intracranial ORR (defined as complete or partial responses in patients with baseline CNS lesions per BICR using RECISTv1.1), intracranial DOR, and PFS. For intracranial assessments, the CNS subgroup was derived per BICR; for systemic analyses, the CNS subgroup was derived per investigator.
4c3880bb027f159e801041b1021e88e8 Result
There were 53 efficacy-evaluable patients with treatment-naïve, ROS1-positive NSCLC. BICR ORR was 77.4% (95% CI 63.8–87.7) with complete responses in three patients (5.7%); median BICR DOR was 24.6 months (95% CI 11.4–34.8). Per baseline CNS status (as determined by investigator), median BICR PFS was 26.3 months (95% CI 15.7–36.6) and 13.6 months (95% CI 4.5–NR) for patients without (n=30) and with CNS disease (n=23), respectively. Intracranial ORR was 55.0% (95% CI 31.5–76.9) and median intracranial DOR was 12.9 months (95% CI 5.6–not reached [NR]) in patients with baseline CNS disease per BICR (n=20). In the overall safety-evaluable population (n=355), most treatment-related AEs were grade 1–2. Few patients required dose reduction (27.3%) or discontinued treatment (3.9%) due to treatment-related AEs.
8eea62084ca7e541d918e823422bd82e Conclusion
Entrectinib was tolerable with a manageable safety profile, and showed clinically meaningful, deep and durable systemic responses in ROS1-positive NSCLC. Clinically meaningful intracranial activity was also demonstrated in patients with baseline CNS disease.
6f8b794f3246b0c1e1780bb4d4d5dc53
Study Sponsor: Ignyta, Inc., a wholly owned subsidiary of F. Hoffmann-La Roche Ltd.Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
OA02.02 - Safety and Preliminary Clinical Activity of Ropotrectinib (TPX-0005), a ROS1/TRK/ALK Inhibitor, in Advanced ROS1 Fusion-Positive NSCLC (ID 14217)
10:40 - 10:50 | Author(s): Alexander Drilon
- Abstract
- Presentation
Background
Ropotrectinib is a potent ROS1/TRK/ALK inhibitor with a >90-fold greater ROS1 potency than crizotinib. Preclinical studies demonstrate robust activity against all known ROS1 resistance mutations, including solvent-front mutation G2032R.
a9ded1e5ce5d75814730bb4caaf49419 Method
In this Phase 1 study (NCT03093116), TKI-naïve and TKI-refractory (≥1 TKI) pts with advanced ALK/ROS1/TRK+ solid tumors received ropotrectinib. Asymptomatic brain metastases were allowed. Primary objectives were to determine MTD and RP2D, with safety, pharmacokinetics, and preliminary antitumor efficacy as the secondary objectives. This is a safety analysis of all pts and subgroup efficacy analysis of the ROS1+ NSCLC pts on the study.
4c3880bb027f159e801041b1021e88e8 Result
As of 16-April-2018, 72 pts have been treated at 6 dose levels from 40mg QD to 200mg BID. Most AEs were grade 1-2. Common (>10%) treatment-related AEs included dizziness (49%), dysgeusia (46%), paresthesias (29%), constipation (19%), fatigue (18%), nausea (11%), and anemia (11%). 4 DLTs were observed at ≥240mg/day: 1 grade 3 (Gr3) dyspnea/hypoxia, 2 Gr3 & 1 Gr2 dizziness. 31 of 72 pts had ROS1+ NSCLC by local testing (FISH, n=20; NGS, n=11) with 1 pt determined as ROS1-negative by central NGS. Antitumor activity in ROS1+ NSCLC has been observed at ROS1 dose levels 40mg QD-160mg BID per investigator assessment, with the best ORR 70% for TKI-naïve and 11% for TKI-refractory pts (17% for 1 prior TKI crizotinib, n=12) (Table). Two crizotinib-resistant pts with G2032R achieved durable cPR and cSD, respectively. Ongoing blinded independent review identified 7 evaluable pts with target CNS lesions at baseline; the intracranial best ORR was 43% (3 cPR, 1 PR*). Updated efficacy data and ctDNA biomarker analyses will be presented.
8eea62084ca7e541d918e823422bd82e ConclusionDose Level
TKI Naïve (n = 10)
TKI Refractory (n = 20)
n
Best Overall Response
n
Best Overall Response
40 mg QD (n = 6)
2
2 cPR (ORR 100%)
4
2 cSD, 1 SD, 1 PD
80 mg QD (n = 5)
2
2 cPR (ORR 100%)
3
1 cSD, 2 SD
160 mg QD (n = 10)
4
2 cPR, 2 cSD (ORR 50%)
6
2 cPR, 2 cSD, 1 SD, 1 PD (ORR 33%)
240 mg QD (n = 2)
1
1 cPR (ORR 100%)
1
1 SD
160 mg BID (n = 7)
1
1 PR*
6
1 PR*, 1 SD*, 1 cSD, 2 SD, 1 NE
Total (n = 30)
10
7 cPR, 1 PR*, 2 cSD
20
2 cPR, 1 PR*, 6 cSD, 1 SD*, 7 SD, 2 PD, 1 NE
Best ORR
70%
11%
Median follow-up
8 months with 90% still on treatment
4 months with 50% still on treatment
cPR: confirmed partial response; SD: stable disease for 2 cycles; cSD: SD for at least 4 cycles; PR* or SD*: waiting for subsequent time point scan; PD: progressive disease; NE: inevaluable; ORR: objective response rate
Ropotrectinib is well tolerated and demonstrates promising activity in pts with advanced ROS1+ NSCLC, including TKI-naïve and TKI-refractory pts. RP2D has not yet been achieved. These Phase 1 data warrant further clinical testing of ropotrectinib in ROS1+ NSCLC.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
OA12 - Novel Therapies in MET, RET and BRAF (ID 921)
- Event: WCLC 2018
- Type: Oral Abstract Session
- Track: Targeted Therapy
- Presentations: 2
- Moderators:
- Coordinates: 9/25/2018, 15:15 - 16:45, Room 106
-
+
OA12.02 - Updated Antitumor Activity of Crizotinib in Patients with MET Exon 14-Altered Advanced Non-Small Cell Lung Cancer (ID 13453)
15:25 - 15:35 | Presenting Author(s): Alexander Drilon
- Abstract
- Presentation
Background
MET exon 14 alterations occur in ~3% of non-squamous non-small cell lung cancer (NSCLCs) and 20–30% of sarcomatoid lung carcinomas. Here we present updated antitumor activity for crizotinib in patients with advanced NSCLC whose tumors are positive for MET exon 14 alterations (hereafter MET exon 14-positive NSCLC), including updated biomarker analyses in circulating tumor DNA (ctDNA).
a9ded1e5ce5d75814730bb4caaf49419 Method
Patients with MET exon 14-positive NSCLC by local molecular profiling were treated with 250 mg crizotinib BID in an expansion cohort of the ongoing PROFILE 1001 study (NCT00585195). Responses were based on derived investigator assessment per RECIST v1.0. Prospective plasma profiling for MET exon 14 alterations in plasma ctDNA was performed (PlasmaSELECT-R64; Personal Genome Diagnostics, Boston, MA).
4c3880bb027f159e801041b1021e88e8 Result
As of Jan 31, 2018, 69 patients (65 response-evaluable) with MET exon 14-positive NSCLC had been treated. Median age was 72 y (range: 34, 91). Tumor histology was: 84% adenocarcinoma, 9% sarcomatoid adenocarcinoma, 4% squamous cell carcinoma and 3% adenosquamous carcinoma. 61% were former-smokers, 38% never-smokers and 1% a current smoker. Median duration of treatment was 7.4 mo (95% CI: 5.5, 9.1), with 29% of patients ongoing. Confirmed responses were 3 CRs and 18 PRs (ORR, 32% [95% CI: 21, 45]); 29 patients had SD as their best overall response (Figure).
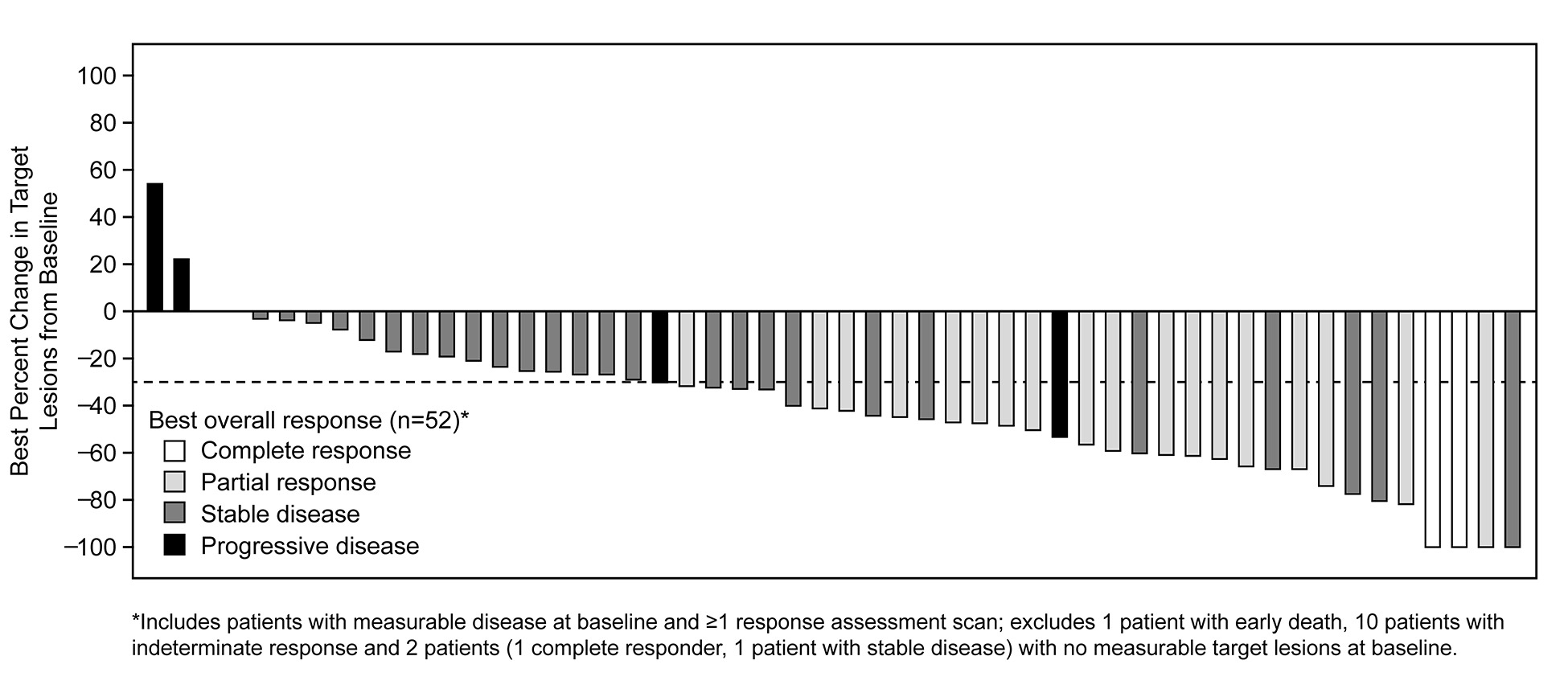
Median time to response was 7.6 weeks (range: 3.7, 16.3). Median DOR was 9.1 mo (95% CI: 6.4, 12.7). Median PFS was 7.3 mo (95% CI: 5.4, 9.1). MET exon 14 alterations were detected in ctDNA from 18/37 (49%) patients with analyzable samples.
8eea62084ca7e541d918e823422bd82e Conclusion
In patients with MET exon 14-positive advanced NSCLC, crizotinib treatment led to objective responses that were rapid and durable, with CRs in some cases. Plasma ctDNA profiling detected MET exon 14 alterations in a subset of patients who harbor MET exon 14 alterations by tumor testing.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
OA12.07 - Clinical Activity of LOXO-292, a Highly Selective RET Inhibitor, in Patients with RET Fusion+ Non-Small Cell Lung Cancer (ID 13922)
16:20 - 16:30 | Author(s): Alexander Drilon
- Abstract
- Presentation
Background
RET kinase gene fusions are actionable drivers that occur in ~2% of non-small cell lung cancers (NSCLC). However, the clinical activity of multikinase inhibitors (MKIs) with anti-RET activity in RET fusion+ NSCLC patients has been limited. LOXO-292 is a highly selective RET inhibitor, with preclinical activity against diverse RET fusions, potential acquired resistance mutations, and against brain metastases.
a9ded1e5ce5d75814730bb4caaf49419 Method
LIBRETTO-001 is a multicenter global phase 1/2 study (26 sites, 9 countries) enrolling patients w/ advanced solid tumors (NCT03157128) including RET fusion+ NSCLC. Patients are dosed orally in 28-day cycles with dose escalation following a 3+3 design. The primary endpoint is MTD/recommended dose determination. Secondary endpoints include safety, overall response rate (ORR, RECIST 1.1) and duration of response (DoR). Initial data were presented at the ASCO 2018 Annual Meeting.
4c3880bb027f159e801041b1021e88e8 Result
As of 02-April 18, 82 solid tumor patients (including 38 RET fusion+ NSCLC) were treated at 8 doses (20 mg QD-240 mg BID). The MTD was not reached. AEs (≥10% of patients) were fatigue (20%), diarrhea (16%), constipation (15%), dry mouth (12%), nausea (12%), and dyspnea (11%); most were grade 1-2. 2 TEAEs ≥ grade 3 were attributed to LOXO-292 (Gr3 tumor lysis syndrome, Gr3 increased ALT). Of the 38 RET fusion+ NSCLC pts, 30 had at least 1 post-baseline assessment or discontinued LOXO-292 prior to such assessment. 26 of 30 patients (87%) had >20% radiographic tumor reduction (range: -21 to -72%). The ORR was 77% (23/30, 3 responses pending confirmation) with a confirmed ORR of 74% (20/27, excluding 3 patients with unconfirmed responses). The response rate was similar regardless of prior MKI treatment (12/15 MKI-naïve, 11/15 MKI pretreated). Responses occurred independent of upstream fusion partner when known (13/16 KIF5B vs 9/11 other) and included patients w/ baseline brain metastases. Most patients remained on treatment (33/38), including all responders. The median DoR was not reached (longest response was the first responder: >10+ months). Rapid plasma clearance of RET variants was observed, with complete clearance by day 15 in 10 of 17 (59%) NSCLC patients with assessable baseline and day 15 ctDNA.
8eea62084ca7e541d918e823422bd82e Conclusion
LOXO-292 was well-tolerated and had marked antitumor activity in RET-fusion+ NSCLC patients, including those w/ resistance to prior MKIs and brain metastases. Phase 2 cohorts are now open globally (160 mg BID). Updated safety and efficacy data as of 19 Jul 2018 will be presented.
6f8b794f3246b0c1e1780bb4d4d5dc53Only Members that have purchased this event or have registered via an access code will be able to view this content. To view this presentation, please login, select "Add to Cart" and proceed to checkout. If you would like to become a member of IASLC, please click here.
-
+
P1.01 - Advanced NSCLC (Not CME Accredited Session) (ID 933)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 2
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.01-74 - MET Exon 14-Altered Lung Cancers: Central Nervous System (CNS) Metastases and Patterns of CNS Progression on MET Inhibition. (ID 14263)
16:45 - 18:00 | Author(s): Alexander Drilon
- Abstract
Background
MET exon 14 (METex14) alterations are targetable drivers found in 3-4% of lung cancers. The frequency of intracranial disease and patterns of central nervous system (CNS) progression on MET tyrosine kinase inhibitors (TKI) are not well characterized.
a9ded1e5ce5d75814730bb4caaf49419 Method
Patients with advanced METex14-altered lung cancers identified by next-generation sequencing (MSK-IMPACT) between January 2014 and March 2018 were eligible for analysis. A retrospective review of clinical features, patterns of metastases, and CNS progression on MET-TKI was performed. The frequency of intracranial disease was compared to cohorts single-center of EGFR-mutant (n=200), ERBB2-mutant (n=98) and KRAS-mutant (n=200) lung cancers.
4c3880bb027f159e801041b1021e88e8 Result
82 patients with metastatic METex14-altered lung cancers were identified. The median age was 73; 56% (n=46) were female and 54% (n=44) were former smokers. The frequency of brain metastases at baseline was 11% (n=9/82). The lifetime frequency of intracranial metastases from diagnosis of metastatic disease was 34% (n=28/82). By comparison, the frequency of brain metastases was 47% (94/200, p=0.05) with EGFR-, 47% (46/98), p=0.09) with ERBB2-, and 32% (64/200, p=0.78) with KRAS-driven tumors. 6% (n=5/82) of patients developed leptomeningeal disease. The overall survival (OS) of patients who developed intracranial disease on therapy compared to those who did not develop intracranial disease was not significantly different (HR 0.66, 95% CI 0.30-1.43, p=0.29). 51 patients received crizotinib, 26 of whom developed progressive disease. The frequency of intracranial (alone), intracranial and extracranial, and extracranial (alone) progression was 8% (2/26), 19% (5/26), and 73% (19/26), respectively.
8eea62084ca7e541d918e823422bd82e Conclusion
A third of patients with METex14-altered lung cancers develop intracranial disease. This proportion is lower than that seen in EGFR- and ERBB2-mutant lung cancers and comparable to KRAS-mutant lung cancers. The frequency of CNS failure on crizotinib was lower than expected compared to historical rates in ALK-rearranged lung cancers.
6f8b794f3246b0c1e1780bb4d4d5dc53 -
+
P1.01-99 - Detecting HER2 Alterations by Next Generation Sequencing (NGS) in Patients with Advanced NSCLC from the United States and China (ID 11285)
16:45 - 18:00 | Author(s): Alexander Drilon
- Abstract
Background
Advances in NGS have led to an increase in identifying specific actionable gene alterations across tumor types. We collected data on HER2 gene alterations detected by NGS from patients with advanced NSCLC and analyzed clinical characteristics and HER2 targeted treatments.
a9ded1e5ce5d75814730bb4caaf49419 Method
Patients diagnosed with advanced NSCLC and underwent NGS testing from Jun 2014 to Dec 2017 at Memorial Sloan-Kettering Cancer Center (MSK) and Guangdong General Hospital (GGH) were included. NGS platforms were MSK-IMPACTTM in MSK and GeneSeek or BurnStone in GGH. Descriptive statistics are used in data analysis.
4c3880bb027f159e801041b1021e88e8 Result
2200 patients from MSK and 490 patients from GGH underwent NGS testing. HER2 mutation and/or amplification were detected in 91/2200(4.1%) patients and 28/490(5.7%) patients from MSK and GGH respectively. Clinical characteristics were listed in Table1. 37.4%(34/91) and 21.4%(6/28) patients from MSK and GGH received HER2 targeted therapies. More patients were enrolled to HER2 inhibitors clinical trials in MSK(24.2%) than GGH(7.1%). The characteristics of HER2 alterations are summarized in Table2.
Table 1. Comparison of HER2 alterations in advanced NSCLC patients from U.S. and China
MSK
N (%)
GGH
N (%)
Total Patients
91
28
Age at Diagnosis (years)
<=60
34 (37.4%)
13 (46.4%)
>60
57 (62.6%)
15 (53.6%)
Sex
Male
37 (40.7%)
14 (50%)
Female
54 (59.3%)
14 (50%)
Smoking History
Former/Current Smoker
53 (58.2%)
7 (25%)
Non-Smoker
38 (41.8%)
21 (75%)
Histology
Adenocarcinoma
84 (92.3%)
25 (89.3%)
Squamous Cell Carcinoma
5 (5.5%)
0
Misc
2 (2.2%)
3 (10.7%)
HER2 status
Mutation
48 (52.7%)
16 (57.1%)
Amplification
32 (35.2%)
11 (39.3%)
Mutation + Amplification
11 (12.1%)
1 (3.6%)
HER2 targeted treatment
34 (37.4%)
6 (21.4%)
Enrolled to HER2 inhibitors clinical trials
22 (24.2%)
2 (7.1%)
8eea62084ca7e541d918e823422bd82e ConclusionTable 2. HER2 alteration in advanced NSCLC patients from U.S. and China combined
NGS Result
Mutation Only
N (%)
Amplification Only
N (%)
Mutation + Amplification
N (%)
Total Patients 64 43 12 Age at Diagnosis (years)
<=60
31 (48.4%)
20 (46.5%)
8 (66.7%)
>60
33 (51.6%)
23 (35.9%)
4 (33.3%)
Sex
Male
39 (60.9%)
19 (44.2%)
7 (58.3%)
Female
25 (39.1%)
24 (55.8%)
5 (41.7%)
Smoking History
Former/Current Smoker
31 (48.4%)
24 (55.8%)
5 (41.7%)
Non-Smoker
33 (51.6%)
19 (44.2%)
7 (58.3%)
Histology
Adenocarcinoma
58 (90.6%)
39 (90.7%)
12 (100%)
Squamous Cell Carcinoma
1 (1.6%)
4 (9.3%)
0
Misc
5 (7.5%)
0
0
HER2 targeted treatment
Yes
19 (29.7%)
14 (32.6%)
7 (58.3%)
No
45 (70.3%)
29 (67.4%)
5 (41.7%)
The incidence and clinical characteristics of HER2 alterations in advanced NSCLC were similar between two large cancer centers in the U.S. and China. These data support U.S.-China collaboration in clinical trials for patients with rare molecular subsets of NSCLC to accelerate new cancer drug development.
6f8b794f3246b0c1e1780bb4d4d5dc53
-
+
P1.13 - Targeted Therapy (Not CME Accredited Session) (ID 945)
- Event: WCLC 2018
- Type: Poster Viewing in the Exhibit Hall
- Track:
- Presentations: 1
- Moderators:
- Coordinates: 9/24/2018, 16:45 - 18:00, Exhibit Hall
-
+
P1.13-40 - Rapid, Robust and Durable Responses to Larotrectinib in Patients with TRK Fusion Non-Small Cell Lung Cancer (ID 14528)
16:45 - 18:00 | Author(s): Alexander Drilon
- Abstract
Background
Neurotrophic tyrosine receptor kinase (NTRK) gene fusions are oncogenic independent of tumor lineage and are widely distributed across cancers. NTRK gene fusions were first reported in lung cancer in 2013 (Vaishnavi et al Nat Med 2013). Larotrectinib is a potent and highly selective oral tropomyosin receptor kinase (TRK) inhibitor in clinical development. Initial data of treatment of 55 patients with TRK fusion cancer resulted in an investigator-assessed objective response rate of 80%, and 71% of patients still in response at one year (Drilon et al., NEJM 2018). We report here on the safety and efficacy of larotrectinib in 4 patients with NSCLC from the 55 patient dataset.
a9ded1e5ce5d75814730bb4caaf49419 Method
Patients with previously treated lung adenocarcinoma were treated under clinical trial and enrolled based on a molecular report of NTRK gene fusion from a CLIA-certified lab. Larotrectinib was administered at 100 mg BID until disease progression or lack of clinical benefit. Tumors were assessed by investigators every 8 weeks using RECIST v1.1 criteria.
4c3880bb027f159e801041b1021e88e8 Result
As of July 17, 2017, four patients with adenocarcinoma of the lung who had progressed after 1 or more lines of platinum-based chemotherapy for advanced disease were enrolled. Three patients harbored an NTRK1 fusion and one an NTRK3 fusion. Three of 4 patients had a partial response or complete response confirmed on a subsequent scan. One patient with a possible brain metastasis demonstrated regression of mass on MRI. Responses were rapid and robust, with a time to response ranging between 49 and 56 days. At the time of analysis, 3 patients continued to have an ongoing response ranging between 5.7 and 12 months. The other patient had stable disease and progressed outside of the CNS after 300 days of treatment and continued on larotrectinib for clinical benefit. Larotrectinib was well tolerated, with 3 of 4 patients having grade 1 events only.
8eea62084ca7e541d918e823422bd82e Conclusion
Larotrectinib treatment resulted in rapid and durable responses and had a well tolerated adverse event profile with no CNS progressive events in patients with previously treated lung cancer harboring NTRK gene fusions. These results strongly support the inclusion of NTRK gene fusions as part of routine testing for patients with lung cancer.
6f8b794f3246b0c1e1780bb4d4d5dc53


